Introduction
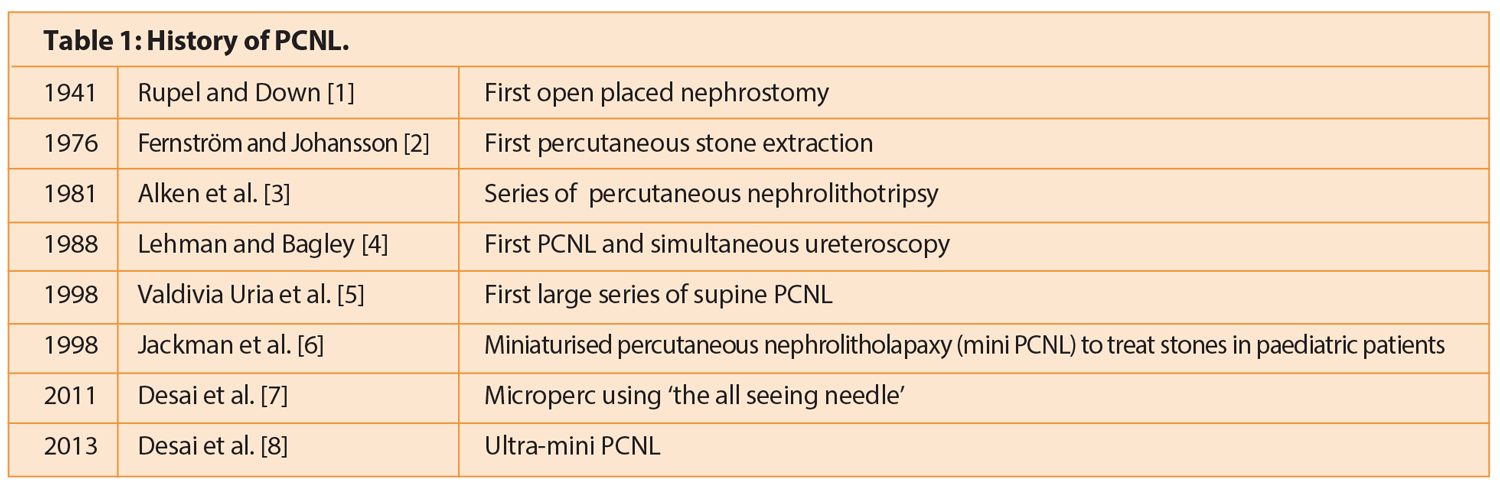
Since the first percutaneous nephrolithotomy (PCNL), the technique has undergone many innovations, including modifications in positioning, miniaturisation of instruments and combination with retrograde intra-renal surgery (see Table 1 for an outline of the history of the technique).
Controversy has emerged as to which is the best position, and / or tract size, for PCNL and a number of variations in patient positioning have been proposed including prone, prone-flexed, lateral, prone split-leg position, total supine and several modified supine positions [9-11]. The recent Clinical research Office of the Endourological Society (CROES) PCNL Global Study data suggests that the majority of PCNL are still performed in the standard prone position and only 20% of all PCNLs are performed supine [12].
Prone versus supine PCNL
The prone position results in a large surface area for kidney puncture (especially important where multiple tracts are required) and a wide space for instrument manipulation. It aids upper pole and anterior calyx access. However, it is associated with anaesthetic risks especially in the morbidly obese or those with compromised cardiopulmonary status. Treatment of morbidly obese patients is becoming a more frequent challenge in urology, as the prevalence of obesity, and associated urolithiasis, is increasing. This highlights the potential benefit of increasing experience with supine PCNL with different anaesthetic risks, better access to the patient’s airway and less repositioning. Supine PCNL also has the potential advantage of greater versatility of intra-renal stone manipulation and can be combined with simultaneous ureteroscopy, although the practicality of this latter point, requiring two competent endourologists and two stacks, is open to question in the NHS.
Theoretically, the angle of the tract may improve removal of stone fragments after lithotripsy and there may be less density overlap with the vertebrae whilst using fluoroscopy. Antero-medial displacement of the kidney during dilation of the tract has been reported in supine PCNL, resulting in a longer, more perpendicular tract [13]. A small but interesting study of 30 patients that underwent reduced dose CT in supine, prone and 30 degree prone-flex positions to identify differences in anatomical variations when gaining percutaneous access revealed mean tract lengths and subcutaneous fat tissue lengths to be significantly longer in the supine position, which may be important in obese patients, resulting in the need to use longer access sheaths [14].
Many of the publications that compare supine and prone PCNL have small sample sizes and are retrospective comparisons. Liu et al. undertook a systematic review and meta-analysis in 2010 which includes two randomised controlled trials and two large prospective case controlled studies. The CROES PCNL Global Study Database report included 5803 patients from 96 international centres, and is the largest collated series found on literature search.
Length of hospital stay
Shoma et al. (n=130) showed no significant difference between length of hospital stay (2.5 days supine vs. 2.7 days prone) [13]. The CROES report found no statistically significant difference in mean hospital stay [12]. Conversely, Mazzucchi et al. (n=56) compared prone versus supine in obese (BMI >30) patients and found that hospital stay was significantly shorter in the supine group – 4.38 days (2-16) prone versus 2.68 days (2-5) supine [15].
Stone-free rates
Liu et al. reported no statistical difference in stone-free rates between prone and supine PCNL (83.5% supine vs. 81.5% prone). De Sio et al. (n=75) compared prone versus supine PCNL in patients with ’uncomplicated stones’ and a BMI <30, finding no significant difference in stone-free rate (88.7% supine vs. 91.6% prone) [17]. Mazzucchi et al. looked at obese patients, also finding no significant difference between final stone-free rates (83.3% prone vs. 78.1% supine) [15]. However, the CROES data found the stone-free rate was significantly higher in prone PCNL (70.2% supine vs. 77% prone) and there were lower rates of failed procedures in prone patients [12]. This was also the case in patients with staghorn calculi (48.4% supine vs. 59.2% prone (P<.001)) [18]. These data are of course international and pooled and have generated much discussion at meetings.

Mean operative time
Liu et al. reported supine PCNL had shorter operative time than prone [16]. Included in that review, De Sio et al. reported a significant difference in mean operative time of 68 minutes prone versus 43 minutes supine, in patients with uncomplicated stones and BMI<30 [17]. Manohar et al. (n=62) reported a mean operative time of 73.66 minutes (range 20-250) in supine PCNL with ultrasound guided access [19]. Mazzucchi et al. reported no significant difference in mean operative time – 120.3 minutes (range 40-300 minutes) prone versus 120.2 minutes (range 30-360 minutes) supine – in obese patients with BMI>30 [15]. Once again varying data from the CROES study found that the mean operative time was lower for prone as compared to supine (82.7 minutes vs. 90.1 minutes), although this is unlikely to be clinically significant [12]. The difference was greater in staghorn calculi (103.2 minutes prone vs. 123.1 minutes supine) [18]. Wang et al. (n=122) also reported a lower mean operative time with prone procedures (78 minutes prone vs. 88 minutes supine (P<0.05)) but felt this might have been affected by the authors’ shorter experience with supine PCNL [20].
Anaesthetic considerations
Prone positioning presents a number of challenges to the anaesthetist. Firstly those related to repositioning, and secondly those related to physiological changes in the prone position. Even safely secured endotracheal tubes can pass into the right main bronchus, secondary to increased neck flexion in prone patients, resulting in lobar collapse. Accidental extubation can occur, especially during repositioning which can be disastrous. Being in the prone position may be more stimulating for the patient, thus requiring a greater depth of anaesthesia, which in combination with decreased cardiac output due to decreased venous return (higher thoracic pressure and / or abdominal compression), can result in the need for more invasive monitoring and potentially cardiovascular support [21].
Should unforeseen complications occur then the insertion of invasive monitoring lines is much more difficult in the prone position [22]. Multiple pressure related injuries have been described secondary to prone positioning including peripheral nerve injuries (brachial plexus, ulnar, radial, lateral cutaneous nerve of the thigh and supraorbital nerve injuries) [23], although these are very rare and tend to happen in patients with specific risk factors (diabetes, peripheral vascular disease, alcohol dependency, pre-existing neuropathy and anatomical variants) [24]. Ocular injuries can include corneal abrasions and globe injury or ischaemia. There are also case reports of hepatic ischaemia after prolonged prone positioning.
Complications
Published transfusion rates after standard PCNL have varied enormously. Keoghane et al. (n=547) reported a transfusion rate of 3.8% in prone PCNL, with one nephrectomy (0.2%) and five selective embolisations (0.9%) [25]. In the first large series of supine PCNLs, Valdivia et al. (n=557) showed a transfusion rate of 1.4%, with one nephrectomy (0.18%) and one selective embolisation (0.18%) [5]. Other reported rates of transfusion in supine PCNL range from 0-9.4% [12,13,15,19,26]. As stone size increases, the transfusion rate increases; CROES data reported transfusion rate of 4.4% in PCNL (all methods) done for single stones 20-40mm, which increased to 13.3% in stones 41-60mm [27]. Liu et al. reported no statistically significant difference in complications, blood transfusion rate or fever, in their systematic review and meta-analysis [16]. The CROES study reported prone patients exhibited higher rates of blood transfusions (6.1% vs. 4.3%) and fever (11.1% vs. 7.6%) [12].
Conversely, Manohar et al. reported 18% postoperative infection in supine PCNL [19]. In one of the few randomised controlled trials, De Sio et al. found no difference in complication rates between the two approaches [17]. One concern urologists have had about supine PCNL is the perceived risk of colonic injury. In fact, in a study of 500 patients who underwent CT scanning prone or supine 2% of patients had a retrorenal colon when supine and increased to 10% when turned prone, therefore the possibility of colonic injury may in fact be lower when supine [28]. Colonic perforation has been reported at 0.2-0.3% in prone PCNL [25-29]. In a review of nine studies for supine PCNL, de la Rosette found 0% reported visceral injuries [30]. CROES suggested overall higher rates of perforation (3.4% supine vs. 3.3% prone) but no difference between the two groups [12].
Learning curve
There are no specific data about the learning curve to achieve surgical competence in supine PCNL but it has been suggested that figures compare to the learning curve for prone PCNL which Tanriverdi et al. suggested was 60 cases (operative time and fluoroscopy screening times drop to steady state after performing 60 procedures) [31] and Ziaee et al. suggested that competence is obtained at 45 cases and excellence after 105 cases [32].
Conclusion
Both prone and modified supine positions appear to be effective and similarly safe, although there may be variations in operative time and stone-free rates dependent on surgeon experience. Supine PCNL should be considered in higher risk anaesthetics, or where there are pre-existing risk factors for pressure-related injuries whereas prone PCNL may be preferable for staghorn calculi.
Miniaturised tracts vs. standard PCNL
In 1998 Jackman et al. developed miniaturised percutaneous nephrolitholapaxy (mini PCNL) to treat stones in paediatric patients [6]. Since then mini PCNL has been utilised to manage adult stones and further innovations have led to the development of the ultra-mini PCNL (UMP) and microperc. The benefits of miniaturised tracts remain controversial; the perceived benefit is that the area of renal parenchyma damaged by a miniaturised tract is decreased compared to standard PCNL, thereby reducing complications. However, the size of the tract also limits flow and fragment extraction methods. Current European Association of Urology (EAU) guidance suggests that stones <2cm in the renal pelvis, middle and upper poles should be approached using extracorporeal shock wave lithotripsy (ESWL), and larger stones should be approached with PCNL [33]. There is an extended indication for PCNL in 1-2cm lower pole stones where there are unfavourable factors for ESWL success, or ESWL has already failed. Although mini PCNL, UMP and microperc are mentioned as a modality to treat renal calculi, it is stated that the benefits remain controversial.
Mini-PCNL
Mini-PCNL is generally reserved for stones <2cm, however some departments have moved solely to mini-PCNL in place of standard PCNL independent of stone size. The role of mini-PCNL in treating a large stone burden remains unclear. The primary stone-free rates using mini-PCNL are higher when operating on smaller stones (<2cm: 90.8% vs. >2cm 76.3%); however secondary stone-free rates are fairly comparable (<2cm: 98.9% vs. >2cm 94.6%) [34]. Stone size will clearly have an impact on operative time when using mini-PCNL. Abdelhafez et al. (n=191) reported a mean operative time of 69.9 minutes in stones <2cm versus 97.4 minutes in stones >2cm, however this was not compared with standard PCNL [34]. For stones between 1-2cm, mean operative time is shorter in standard PCNL. Misra et al. (n=55) reported that in mini-PCNL mean operative times in 1-2cm stones were longer (mini-PCNL: 45.2 minutes vs. standard PCNL: 31 minutes) [37]; Knoll et al. (n=50) also showed a tendency (non-significant) towards longer operative times in mini PCNL patients (mean stone size = 18mm, mean operative time = 59 minutes) in comparison to standard PCNL (mean stone size = 22mm, mean operative time = 49 minutes) with uncomplicated solitary renal calculi [38].
The prolonged operative time may be related to the need for more extensive stone fragmentation. Screening times are comparable. Abdelhafez investigated the use of mini-PCNL in stones >2cm (mean 36.7mm), separating them into complex and simple stones with comparable screening times (complex: 210 seconds, simple: 222.0 seconds) [39]. Standard PCNL fluoroscopic screening times can vary from 96 to 611 seconds and are longer with larger stone burden or multiple tracts [40,41]. Knoll showed that tubeless mini-PCNL could decrease length of stay by 0.9 day (mean stay = 3.8 days standard PCNL versus 2.9 tubeless mini PCNL) [38].
Complication rates of mini-PCNL of 26.5% were reported by Abdelhafez. Of these, the majority were Clavien 1 (77%) and no grade 4 or 5 complications occurred in this group [39]. This is similar to CROES data. Of 5724 PCNL patients 20.5% experienced one or more complications, the majority of which (54%) were Clavien grade 1 [42]. As suspected, several studies report lower transfusion rate in mini-PCNL compared to standard PCNL. Abdelhafez reported transfusion rate of 1.2% in mini-PCNL [39] whilst Wang et al. reported a 1.8% transfusion rate in mini-PCNL versus 7.2% standard PCNL [43]. Zimmermann et al. presented a series of 650 mini-PCNL cases, reporting a transfusion rate of 1.4% with two cases of embolisation [44]. The difference may be less marked when dealing with larger calculi; the haemoglobin drop in <2cm stones was 1.3g/dL compared to 1.7g/dL in >2cm stones in mini-PCNL (P=0.015) [34]. As with supine PCNL, there are limited data on the learning curve for mini-PCNL – what we do know is that it takes more than 35 cases to gain competence for a novice [45].
Microperc
Bader et al. presented their experience using the ‘all-seeing needle’ to confirm the quality of the access prior to dilatation of the operating tract [46]. This was then developed further and Desai et al. reported using this 4.85Fr needle to perform a single step microperc to treat renal calculi [7]. The proposed area of use, although not specified in the current EAU guidelines, is as an alternative in patients with moderate stone burden (1-2cm) [47]. With regards to length of stay in hospital, microperc patients stay for between one and three days [47], with one study of 140 renal units stating a mean of 1.76 days [36].
When examining stone-free rates with microperc there has been variable success. Campobasso et al. looked at multiple series of microperc patients. They identified a total of 196 treated renal units showing a primary stone-free rate of between 82.14% and 97.7% [47]. Hatipoglu (n=140) showed comparable stone-free rate across a variety of sizes of stone (≤10mm: 87.1, 10-20mm: 83.8% and ≥20mm 87.1%) [36]. It is important to bear in mind paucity of the data when examining the current evidence for microperc. Operative times with microperc have been reported between 55.7 minutes (mean stone size = 15.0mm) to 63.5 minutes (mean stone size = 17.9mm) [36,48]. With regards to fluoroscopic screening times and microperc means vary from 107.4 (mean stone size = 15.07mm) to 150.5 seconds (mean stone size = 17.8mm) [36,49]. Hatipoglu et al. also reported a total of 20 complications (14.28%) in patients treated with microperc – the majority (55%) were Clavien grade 1 (35%) or 2 (20%) [36]. A very low transfusion rate was shown in microperc patients of 1/140 (0.7%) [36].
With all the potential benefits of microperc it is also important to bear in mind some of the other potential flaws. The smaller tract can lead to the inability to retrieve all fragments, which can lead to an increase in the percentage of patients requiring ureteric stenting [47]. The smaller tract may also lead to an increase in intra renal pelvic pressure and this pressure leads to a greater chance of transient bacteremia and subsequent sepsis [50]. Overall the initial data for microperc seems promising. The low complication rates and high success rates that have been demonstrated are to be balanced against a smaller tract but preferably more randomised data are needed.
FURS versus PCNL
Stones <2cm in the renal pelvis have been traditionally approached with ESWL and then flexible ureterorenoscopy (FURS), with miniaturised tracts now joining the armamentarium. Percutaneous access may have definite advantages over FURS to access calyceal diverticulae or lower calyces with acute infundibular angles, and appear otherwise comparable except for hospital stay. When comparing mini-PCNL and FURS we can see that there are certain advantages to mini-PCNL. In a small prospective but non-randomised study, Knoll (n=46) showed that operative time, number of procedures and immediate stone-free rates were lower in a mini-PCNL group in comparison to FURS for comparable stone sizes (mini-PCNL mean: 18mm, FURS mean: 19mm). There were no major complications in either group and Clavien I and II complications were surprisingly lower, although non-significant, in the mini-PCNL group (mini-PCNL: 16%, FURS: 23.8% (P=0.13)) [51].
In one randomised controlled trial (n=70) comparing microperc to FURS for stones <1.5cm, operative times and stone clearance rates were comparable (microperc 97.1% vs. FURS 94.1%). There was a higher requirement for postoperative stenting in the FURS group, where as microperc was associated with a higher haemoglobin loss and increased pain and analgesic requirements [52]. Bozkurt et al. (n=79) compared standard PCNL to retrograde intrarenal surgery (RIRS) in lower pole stones of 15-20mm, finding that the primary stone-free rate was not significantly different (92.8% PCNL vs. 89.2% RIRS), complication rate was comparable, operative time was significantly higher in RIRS and hospital stay was longer in the PCNL group [53].
Historically, 2-3cm stones would have been tackled by PCNL, but RIRS is now considered a viable alternative for selected patients, although with the caveat that primary stone-free rates are lower (Giusti: mean stone size 2.7cm, primary stone-free rate RIRS 66% [54]; Akman: stone size 2-4cm, primary stone-free rate 73.5% RIRS vs. 91.2% PCNL [55]), operative times are longer (58.2 minutes RIRS vs. 38.7 minutes PCNL [55]) and multiple sessions (usually day-cases) are likely to be required.
Conclusion
The exact role for smaller tracts has not yet been ascertained (in keeping with recent EAU guidelines). But what the limited research presented here alludes to, is a possible, future role for microperc and mini-PCNL. The exact application of these tools will only come with further more substantial research including randomised controlled trials but it would seem to lie in the treatment of stones <2cm. As stone surgery progresses, and with the advent and application of stone multidisciplinary meetings, stone units will be able to apply the most appropriate technique for any particular stone, be it an endoscopic or percutaneous approach, small or standard tract or supine versus prone approach.
References
1. Rupel E, Brown R. Nephroscopy with removal of stone following nephrostomy for obstructive calculus anuria. J Urol 1941;46:177-82.
2. Fernstrom I, Johansson B. Percutaneous nephrolithotomy: A new extraction technique. Scand J Urol Nephrol 1976;10:257.
3. Alken P, Hutschenreiter G, Gunther R, Marberger M. Percutaneous stone manipulation. Urology 1981;125:463-6.
4. Lehman T, Bagley D. Reversed lithotomy: modified prone position for simultaneous nephroscopic and ureteroscopic procedures in women. Urology 1988;32:529-32.
5. Valdivia Uria JG, Valle Gerhold J, Lopez Lopez JA, et al. Technique and complications of percutaneous nephroscopy: experience with 557 patients in the supine position. J Urol 1998;160:1975-8.
6. Jackman SV, Docimo SG, Cadeddu JA, et al. The “mini-perc” technique: a less invasive alternative to percutaneous nephrolithotomy. World Journal of Urology 1998;16(6):371-4.
7. Desai MR, Sharma R, Mishra S, et al. Single-Step Percutaneous Nephrolithotomy (Microperc): The Initial Clinical Report. J Urol 2011;186:140-5.
8. Desai J, Solanki R. Ultra-mini percutaneous nephrolithotomy (UMP): one more armamentarium. BJU international 2013;112(7):1046-9.
9. Daels F, Gonsalez MS, Freire FG, et al. Percutaneous Lithotripsy in Valdivia-Galdakao decubitus position: Our experience. J Endourol 2009;23:1615-20.
10. McCahy P, Rzetelski-West K, Gleeson J. Complete stone clearance using a modified supine position: initial experience and comparison with prone percutaneous nephrolithotomy. J Endourol 2013;27:705-9.
11. Kumar P, Bach C, Kachrilas S, et al. Supine percutaneous nephrolithomy (PCNL) ‘in vogue’ but in which position? BJU international 2012;110:E1018-21.
12. Valdivia JG, Scarpa RM, Duvdevani M, et al. Supine versus Prone Position during percutaneous nephrolithotomy: A Report from the Clinical Research Office of the Endourological society percutaneous nephrolithotomy Global Study. J Endourol 2011;25(10):1619-25.
13. Shoma A, Eraky I, El-kenawy M, El-Kappany H. Percutaneous Nephrolithotomy in the supine position: technical aspects and functional outcome compared with the prone technique. Urology 2002;60:388-92.
14. Yazici CM, Kayhan A, Dogan C. Supine or prone percutaneous nephrolithotomy: Do anatomical changes make it worse? J Endourol 2014;28(1):10-16.
15. Mazzucchi E, Vicentini FC, Maarchini GS, et al. Percutaneous nephrolithotomy in Obese patients: Comparison between the prone and total supine position. J Endourol 2012;26(11):1437-42.
16. Liu L, Zheng S, Xu Y, Wei Q. Systematic review and Meta-analysis of Percutaneous Nephrolithomy for patients in the Supine Versus Prone Position. J Endourol 2010;24(12):1941-6.
17. De Sio M, Autorino R, Quarto G, et al. Modified supine versus prone position in percutaneous nephrolithotomy for renal stones treatable with a single percutaneous access: a prospective randomized trial. Eur Urol 2008;54:196-203.
18. Astroza G, Lipkin M, Neisius A, et al. on behalf of the CROES PNL study group. Effect of Supine vs. Prone Position on Outcomes of Percutaneous Nephrolithotomy in Staghorn Calculi: Results from the Clinical Research Office of the Endourology Society Study. Urology 2013;82:1240-5.
19. Manohar T, Jain P, Desai M. Supine Percutaneous Nephrolithotomy: Effective Approach to High-risk and Morbidly obese patients. J Endourol 2007;21(1):44-9.
20. Wang Y, Wang Y, Yao Y, et al. Prone versus modified supine position in percutaneous nephrolithotomy: a prospective randomized study. Int J Med Sci 2013;10:1518-23.
21. Atkinson CJ, Turney BW, Noble JG, et al. Supine vs. Prone Percutaneous Nephrolithotomy: An Anaesthetist’s view. BJU International 2011;108(3):306-8.
22. Sunder-Plassmann G, Locker GJ, Muhm M, et al. Central venous catheterization in a patient in the prone position. Crit Care Med 1997;25:1439-40.
23. Edgecombe H, Carter K, Yarrow S. Anaesthesia in the prone position. Br J Anaesth 2008;100:165-83.
24. Winfree CJ, Kline DG. Intraoperative positioning nerve injuries. Surg Neurol 2005;63:5-18.
25. Keoghane SR, Cetti RJ, Rogers AE, Walmsley BH. Blood transfusion, embolisation and nephrectomy after percutaneous nephrolithotomy (PCNL). BJU International 2012;111:628-32.
26. Rana AM, Bhojwani JP, Junejo NN, Bhagia SD. Tubeless PCNL with Patient in Supine Position: procedure for all seasons?- with comprehensive technique. Urology 2008;71:581-5.
27. Xue W, Pacik D, Boellaard W, et al. on behalf of the CROES PCNL group. Management of Single Large Nonstaghorn Renal stones in the CROES PCNL Global Study. J Urol 2012;187(4):1293-7.
28. Hopper KD, Sherman JL, Luethke JM, et al. The retrorenal colon in the supine and prone patient. Radiology 1987;162:443-6.
29. El-Naha AR, Shokier AA, El-Assmy AM, et al. Colonic perforation during percutaneous nephrolithomy: study of risk factors. Urology 2006;67:937-41.
30. De la Rosette JJ, Tsakiris P, Ferradino MN, et al. Beyond prone position in percutaneous nephrolithotomy: a comprehensive review. Eur Urol 2008;54:1262–9.
31. Tanriverdi O, Boylu U, Kendirci M, et al. The learning curve in the training of percutaneous nephrolithotomy. Eur Urol 2007;52:199-204.
32. Ziaee SAM, Sichani MM, Kashi AH, Samzadeh M. Evaluation of the learning curve for percutaneous nephrolithotomy. Urology 2010;7(4):226-31.
33. Turk C, Knoll T, Petrik A, et al. EAU Guidelines on Urolithiasis. Chapter 6: Indication for active stone remoal and selection of procedure. 2014:42-51.
34. Abdelhafez MF, Amend B, Bedke J, et al. Minimally invasive percutaneous nephrolithotomy: a comparative study of the management of small and large renal stones. Urology 2013;81(2):241-5.
35. Desai J, Zeng G, Zhao Z, et al. A novel technique of ultra-mini-percutaneous nephrolithotomy: introduction and an initial experience for treatment of upper urinary calculi less than 2cm. Biomed Res Int 2013; Epub ahead of print.
36. Hatipoglu NK, Tepeler A, Buldu I, et al. Initial experience of micro-percutaneous nephrolithotomy in the treatment of renal calculi in 140 renal units. Urolithiasis 2014;42(2):159-64.
37. Mishra S, Sharma R, Garg C, et al. Prospective comparative study of miniperc and standard PNL for treatment of 1 to 2 cm size renal stone. BJU International 2011;108(6):896-900.
38. Knoll T, Wezel F, Michel MS, et al. Do patients benefit from miniaturized tubeless percutaneous nephrolithotomy? A comparative prospective study. J Endourol 2010;24(7):1075-9.
39. Abdelhafez MF, Bedke J, Amend B, et al. Minimally invasive percutaneous nephrolitholapaxy (PCNL) as an effective and safe procedure for large renal stones. BJU International 2012;110(11 pt C):E1022-6.
40. Keoghane SR, Cetti RJ, Hanna L, et al. Minimising radiation exposure during percutaneous nephrolithotomy (PCNL) - A series of 348 patients. SIU UK Section Meeting (London, UK; November 22, 2013).
41. Tepeler A, Binbay M, Yuruk E, et al. Factors affecting the fluoroscopic screening time during percutaneous nephrolithotomy. J Endourol 2009;23(11):1825-9.
42. Labate G, Modi P, Timoney A, et al. on behalf of the CROES PCNL Study Group. The percutaneous nephrolithotomy global study: classification of complications. J Endourol 2011;25(8):1275-80.
43. Wang XQ, Wang CX, Hao YY. The comparison of minimally invasive tract and standard tract in percutaneous nephrolithotomy for renal stones. Eur Urol Suppl 2011;10(2):70-1.
44. Zimmermann V, Liske P, Lahme S. Minimally invasive PCNL (MPCNL) -proven efficiency and safety after more than 650 consecutive patients. J Urol 2011;185(Suppl.):e772.
45. Schilling D, Gakis G, Walcher U, et al. The learning curve in minimally invasive percutaneous nephrolitholapaxy: a 1-year retrospective evaluation of a novice and an expert. World Journal of Urology 2011;29(6):749-53.
46. Bader MJ, Gratzke C, Seitz M, et al. The “all seeing needle”: initial results of an optical puncture system confirming access in percutaneous nephrolithotomy. Eur Urol 2011;59(6):1054-9.
47. Campobasso D, Ferretti. Is Microperc really the new frontier of percutaneous nephrolithotripsy? Urolithiasis 2014; Epub ahead of print.
48. Armagan A, Tepeler A, Silay MS, et al. Micropercutaneous nephrolithotomy in the treatment of moderate-size renal calculi. J Endourol 2013;27(2):177-81.
49. Tepeler A, Armagan A, Sancaktutar AA, et al. The role of microperc in the treatment of symptomatic lower pole renal calculi. J Endourol 2013;27(1):13-18.
50. Tepeler A, Akman T, Silay MS, et al. Comparison of intrarenal pelvic pressure during micro-percutaneous nephrolithotomy and conventional percutaneous nephrolithotomy. Urolithiasis 2014;42(3):275-9.
51. Knoll T, Jessen JP, Honeck P, Wendt-Nordahl G. Flexible ureterorenoscopy versus miniaturized PNL for solitary renal calculi of 10–30 mm size. World Journal of Urology 2011;29(6):755-9.
52. Sabnis RB, Ganesamoni R, Doshi A, et al. Micropercutaneous nephrolithotomy (microperc) vs retrograde intrarenal surgery for the management of small renal calculi: a randomized controlled trial. BJU International 2013;112(3):355-61.
53. Bozkurt OF, Resorlu B, Yildiz Y, et al. Retrograde intrarenal surgery versus percutaneous nephrolithotomy in the management of lower-pole renal stones with a siameter of 15-20mm. J Endourol 2011;25(7):1131-5.
54. Giusti G, Proietti S, Luciani LG, et al. Is retrograde intrarenal surgery for the treatment of renal stones with diameters exceeding 2cm still a hazard? Can J Urol 2014;21(2):7207-12.
55. Akman T, Binbay M, Ozgor F, et al. Comparison of percutaneous nephrolithotomy and retrograde flexible nephrolithotripsy for the mangement of 2-4cm stones: a matched pair analysis. BJU International 2012;109(9):1384-9.







