Chronic testicular pain (CTP) is defined as constant or intermittent, unilateral or bilateral testicular pain of more than three months’ duration, which significantly interferes with the daily activities of the patient prompting medical advice [1-4].
This condition is commonly seen by urologists and is difficult to treat. It is often frustrating, both for urologists and patients, because of multiple aetiological factors which contribute to it, and the absence of a standardised, widely accepted treatment algorithm and care [5]. In almost every case there is a need for bespoke treatment. There are 100,000 cases of chronic testicular pain every year in the USA and the incidence is increasing because of rising health awareness [6].
Chronic testicular pain is one of the commonest reasons for medical discharge from the US army [7]. In the majority of cases pain is idiopathic (up to 50%), while in the rest pain is related to previous infection or trauma [1,5]. Post vasectomy pain syndrome is documented in up to 19% of men undergoing vasectomy [8]. The BAUS vasectomy information leaflet quotes a 5-30% risk of chronic testicular pain [9].
There are several medical and surgical treatments recommended in the published literature for CTP. In our unit a senior consultant urologist with a special interest in the management of CTP looks after all referrals with this condition, including regional referrals. At least 20% of referrals are from regional centres. We believe that every patient with CTP should be thoroughly assessed, investigated and offered treatment depending on the clinical findings and aetiological factors.
In this feature article, we discuss the surgical management of CTP and report the outcome data for procedures performed by a single urologist over a five-year period.
Patient evaluation
A thorough history is crucial in the assessment of the patient, including the relevant medical, sexual and surgical history. It’s important at this stage to rule out non-scrotal aetiological factors causing referred pain from musculoskeletal or intra-abdominal pathology (e.g. distal ureteric stone). It is important to ask for duration, nature and any trigger factors for the pain.
Physical examination of the groin and genitals is undertaken, as well as examination of the abdomen. Scrotal examination may pinpoint the painful area, and digital rectal examination is undertaken to evaluate pelvic floor and prostate. We recommend scrotal examination both in laying and standing position, in particular to elicit signs suggestive of varicocele, or retraction of testis in the standing position indicating hyperactive cremaster muscle.
“This condition is often frustrating, both for urologists and patients, because of multiple aetiological factors which contribute to it, and the absence of a standardised, widely accepted treatment algorithm and care.”
Urine examination and an ultrasound examination of testes are first-line investigations. CT scan of abdomen and MRI of spine are also considered, depending on the clinical suspicion of the source of pain. Increased intravesical pressure may be associated with testicular pain as well [10].
Differential diagnosis
These include chronic epididymitis, post-vasectomy pain syndrome, intermittent torsion, hyperactive cremaster muscle, varicocele, and referred pain secondary to interstitial cystitis, chronic prostatitis, distal ureteric stone, and intervertebral disc prolapse.
Surgical management
Failed medical management with analgesia and a surgically motivated patient prompts surgical intervention. Almost all patients have been on fairly strong analgesics prior to referral to us, including co-codamol, pregablin, gabapentin and amitriptyline. We have used cord block (local anaesthetic + steroids) in a number of patients prior to advising for stripping of the cord with varied results. It does not always predict if the surgery will be successful.
Figure 1.
Microsurgical spermatic cord nerve stripping
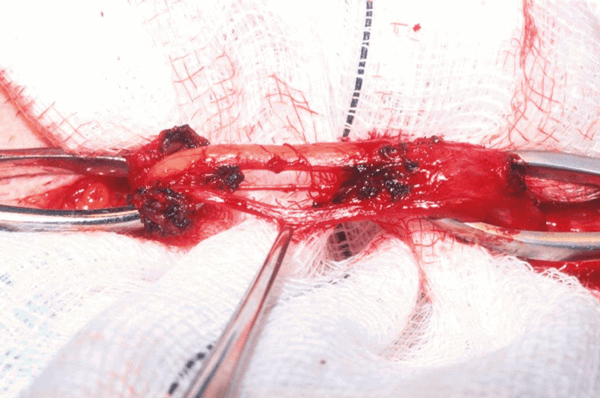
This procedure is advised in patients with post vasectomy and idiopathic chronic testicular pain. It involves inguinal incision and microsurgical dissection of the spermatic cord with preservation of only the testicular artery, vas deferens and vein [Figure 1]. Use of an operating microscope is usually recommended. However, we use surgical magnifying loops with equally good results. Risks associated with this procedure, which patients are counselled for, include testicular atrophy, hydrocele, infection and haematoma.
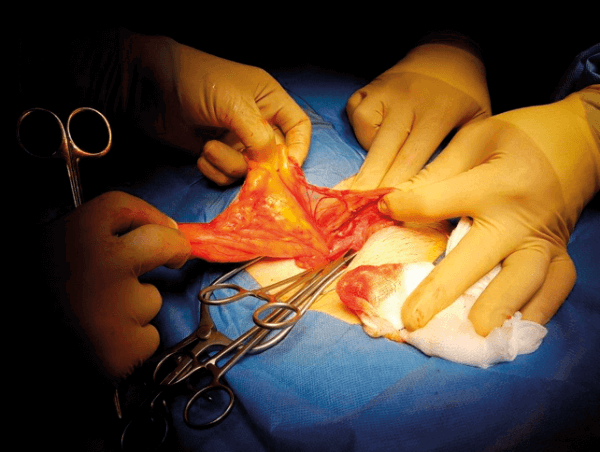
This procedure is believed to work through destruction of degenerated cord nerves that are found in the cremaster muscle fibres, spermatic fascia, and in relation to the vas deferense and the internal spermatic arteries [11]. It is successful in 60-90% of cases [12]. Infrequently a large lipoma is encountered during exploration of the spermatic cord [Figure 2 and 3]. We have observed it in 20% of cases. This is excised. We are uncertain about the significance of the presence of lipoma of the cord and effects of its removal on final outcome.
Figure 2.
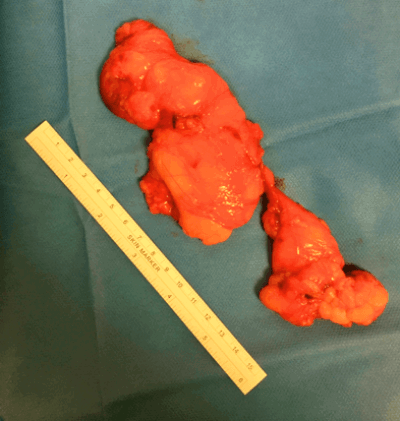
Figure 3.
Division of cremaster muscle
Cremaster muscle division is recommended in patients with hyperactive cremaster muscle. They have very mobile testicles, ascending to the groin with physical activity and causing pain. Pain settles down once testis is manually brought down to the scrotum. This procedure is performed through inguinal incision and complete division and ligation of cremaster is done. This procedure is very safe with minimal risk of infection and haematoma.
Bilateral testicular fixation
Intermittent testicular torsion typically presents as testicular pain, which is self-limiting and lasts for few minutes to few hours, in a younger population. Completely normal physical examination and normal ultrasound scan would help to exclude any other pathology. Procedure is performed through a midline scrotal incision, and bilateral three-point fixation is done with 2/0 Vicryl or PDS.
Vasectomy reversal
Vasectomy reversal is an option for post vasectomy chronic pain syndrome when all other options fail. This procedure is recommended for patients with epididymal congestion type dull pain and pain after ejaculation. On examination the epididymis may be tender and feels bulky. We perform end-to-end vaso-vasostomy with 7/0 PDS in two layers. This procedure is not very popular with patients because of reversal of fertility. This procedure can also be offered after failure of spermatic cord nerve stripping. The published evidence suggest a 75-90% success rate [13].
“A thorough history is crucial in the assessment of the patient, including the relevant medical, sexual and surgical history.”
Orchidectomy
Extirpative surgery is recommended as a last resort in the published literature. In unilateral conditions it may be considered after failed medical and surgical treatments. In the case of bilateral pain, before offering orchiectomy it is advised to take a multidisciplinary approach. A thorough psychosomatic assessment and chronic pain team input is sought. We advise to avoid orchidectomy whenever possible. Documented improvement in pain varies between 20% and 70% [14]. There is also a 25% association with ‘phantom testicular pains’ [15]. We have not done any orchidectomy in our unit for CTP.
Our data over last five years
We have prospectively collected our outcomes data from January 2013 in a handwritten diary maintained by the team. Preoperative findings and differential diagnosis are recorded. The operative procedure and operative surgeon data are also recorded, as are the outcomes from patient review in the follow-up clinic (Table 1).
Table 1.
Procedure: Spermatic cord nerve stripping
Number of patients: 35
% response: 91%
Procedure: Cremaster muscle division
Number of patients: 21
% response: 100%
Procedure: Bilateral testicular fixation
Number of patients: 9
% response: 100%
Procedure: Vasectomy reversal
Number of patients: 3
% response: 100%
Procedure: Epidydimo-vasostomy
Number of patients: 1
% response: 100%
Total
Number of patients: 69
% response: 99%
A total of 69 patients were operated on for chronic testicular pain in our unit. All operations were either performed or supervised by a single consultant urological surgeon (Mr Naeemuddin Shaikh). The surgical procedures performed on these 69 patients were: spermatic cord nerve stripping – 35 (50.72%), cremaster muscle division – 21 (30.43%), bilateral testicular fixation – 9 (13%), vasectomy reversal – 3 (4.34%), and epidydimo-vasostomy – 1 (1.45%). All these patients were followed up in clinic or had telephonic consultation (due to travelling distance for regional referrals) after three months. Surgical outcomes were recorded based on improvement in pain and postoperative complications. Pain improvement was assessed based on numerical rating scale (NRS 0-10).
We have not encountered any significant postoperative complications apart from a small haematoma and minor wound infection in one case. More than 90% of patients had a good improvement in pain and quality of life (score ≤2). Seven percent of patients had 50% improvement in their symptoms (score ≤5) and they were happy with the outcome, while 3% of nerve stripping procedures had no response.
“Provided care is taken to make a correct diagnosis and an appropriate surgical procedure is carried out then the outcome can be excellent.”
Summary
There is a clear role for surgery in patients who have had no response to a conservative approach. The management of CTP is challenging and decisions on the appropriate operative procedure is largely based upon a thorough history and examination. Provided care is taken to make a correct diagnosis and an appropriate surgical procedure is carried out then the outcome can be excellent. The surgical options should be offered to patients if a correct diagnosis is made at the outset as the outcomes may be rewarding.
References
1. Davis BE, Noble MJ, Weigel JW, et al. Analysis and management of chronic testicular pain. J Urol 1990;143:936-9.
2. Tan WP, Levine LA. What can we do for chronic scrotal content pain? World J Men Health 2017;35(3):146-55.
3. Tan WP, Levine LA. An overview of the management of post-vasectomy pain syndrome. Asian Journal of Andrology 2016;18:332-7.
4. Tan WP, Tsambarlis PN, Levine LA. Microdenervation of the spermatic cord for post‐vasectomy pain syndrome. BJUI 2018;121(4):667-73.
5. Levine LA. Chronic orchialgia: evaluation and discussion of treatment options. Ther Adv Urol 2010;2(5-6):209-14.
6. Calixte N, Brahmbhatt J, Parekattil S. Genital pain: algorithm of management. Transl Androl Urol 2017;6(2):252-7.
7. Kavoussi PK, Costabile RA. Orchialgia and the chronic pelvic pain syndrome. World J Urol 2013;31:773-8.
8. Ahmed I, Rasheed S, White C, Shaikh NA. The incidence of post-vasectomy chronic testicular pain and the role of nerve stripping (denervation) of the spermatic cord in its management. Br J Urol 1997;79(2):269-70.
9. Shaikh NA, Blacklock ARE, Arkell DG. Urodynamic assessment of patients with previous epididymo-orchitis and chronic testicular pain. Proceedings of AUA Annual Meeting, San Francisco, 1994.
10. Parekattil SJ, Priola KB, Atalah HN, et al. Trifecta of pain: anatomic basis for denervation of the spermatic cord for chronic orchialgia. J Urol 2010;183(Suppl):e730-e731.
11. Larsen SM, Benson JS, Levine LA. Microdenervation of the spermatic cord for chronic scrotal content pain: single institution review analyzing success rate after prior attempts at surgical correction. J Urol 2013;189:554-8.
12. Werthman P. Vasectomy reversal for post-vasectomy pain syndrome: a ten-year experience. J Urol 2010;183(Suppl):e752.
13. Lowe G. Extirpative surgery for chronic orchialgia: is there a role? Transl Androl Urol 2017;6(Suppl 1):S2-S5.
14. Pühse G, Wachsmuth JU, Kemper S, et al. Phantom testis syndrome: prevalence, phenomenology and putative mechanisms. Int J Androl 2010;33(1):e216-20.
Declaration of competing interests: None declared.