Gender incongruence arises when there is a mismatch between an individual’s gender identity and their sex assigned at birth. Genital gender affirmation surgery (GAS) is the final step of transition for transgender and non-binary individuals who experience gender incongruence. This transition involves a series of surgical procedures that can be individually adapted to each patient to further align their physical appearance with their gender identity.
Genital GAS for individuals assigned female at birth is commissioned at several centres in the UK. The only fully operational specialist unit is based at New Victoria Hospital, Kingston for NHS-funded patients and St Peter’s Andrology Centre, London in the independent sector. The surgery is complex and offered in several stages to allow for recovery and reassessment between each stage.
The aim of this article is to promote some understanding of the surgical procedures involved for transmasculine genital GAS and associated complications. Such an understanding is important because the urologist on call may be referred an individual who has presented following genital GAS with a complication. Furthermore, the rapidly increasing number of individuals who have completed one or more stages of genital GAS will mean that a urologist may be faced with managing unrelated urological issues in these individuals.
Genital GAS options
The options for transmasculine genital GAS are either a metoidioplasty or a phalloplasty performed as a ‘first’ stage. Metoidioplasty is the lengthening of the clitoris to form a small penis after at least two years of gender affirmation hormone therapy (testosterone) [1]. Phalloplasty is the creation of a full-sized neophallus from a free or pedicled flap. To complete their transition the patient may, and often does, opt for adjunct procedures such as glans sculpting, vaginectomy, clitoral transposition, scrotoplasty and insertion of an erectile device and testicular prosthesis. In the UK, these procedures are completed in two further stages. Hence, a complete transition will take approximately 12-18 months. These patients have a 94-100% satisfaction rate with their final results [2].
Acute presentations following genital GAS
Haematoma and bleeding
The sequalae of developing a haematoma is highest following phalloplasty. This is because a haematoma may cause compression of the vascular pedicle, which could lead to ischaemia and partial (or total) loss of the phallus. Patients are inpatient for a week following phalloplasty for close monitoring. The risk of bleeding is lower following discharge but if found, the specialist unit should be contacted immediately for advice and transfer. A haematoma can also drain to the neo-scrotum after the second stage (usually due to vaginectomy) or the third stage (insertion of erectile device and testicular prosthesis). Small haematomas are often treated conservatively but larger haematomas may require surgical evacuation. A CT angiogram may be necessary to assess severity and to localise active bleeding prior to radiological embolisation or surgical exploration.
Suprapubic catheter issues
Postoperatively the suprapubic catheter (SPC) is kept for three weeks to divert the urine. If the catheter falls out early, a new one should be re-sited. This should be done under ultrasound and direct cystoscopic control to avoid injuring the vascular pedicle of the phalloplasty (or abdominal contents). Alternatively, retrograde insertion of a urinary catheter may be considered after discussion with the specialist unit. Voiding per urethra too early may cause a urethral fistula.
Infection and sepsis
The erectile device infection rate is 8.5% following phalloplasty with the majority of these occurring during the first month [3]. Broad-spectrum intravenous antibiotic therapy should be started in addition to adequate resuscitation if an infected device is suspected. The specialist unit should be contacted to arrange urgent transfer for surgical removal of the implant. Explant is often challenging due to the polyethylene terephthalate (Dacron®, Getinge, UK) graft and suture material used to anchor the device. All components including the polyethylene terephthalate cap within the tip of the phallus and all suture material should be removed. Otherwise, they will form a nidus for future infection.
Long-term complications
Erectile device complications
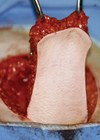
Implantation of an erectile device is part of the final stage of transition if patients desire to have penetrative sexual intercourse. The management of complications remains the same as for any patient with an erectile device. If there is an impending erosion but the device is not externalised, close surveillance of the skin should be performed to check for any skin breaks and to start antibiotics if appropriate. Though the patient will need surgery, it can be done as an urgent elective procedure. Therefore, the patient doesn’t always need transferring to their specialist unit, though a phone referral to allow an urgent clinic or elective admission within one to two weeks is advisable. However, if the device is visible through the skin or urethra (Figure 1), this is a surgical emergency and the device should be removed on that admission.

Figure 1: Erectile device eroded through tip of phallus.
Antibiotic therapy should be started as soon as possible prior to transferring the patient to the specialist unit for removal. Due to the complex anatomy following penile reconstruction, we do not recommend general urologists remove these implants.
All inflatable erectile devices have a life span of approximately 10 years depending on the use by the patient. Early mechanical failure can occur in 30% of cases with one study putting the five-year device survival at 78% [3]. The patient should be referred back to their specialist unit for revision.
Urethral strictures and fistulas
Urethral complications including strictures and fistulas are the most common complications following genital GAS. In our centre there is a 43.5% urethral complication rate after a median follow-up of 3.9 years [4,5].
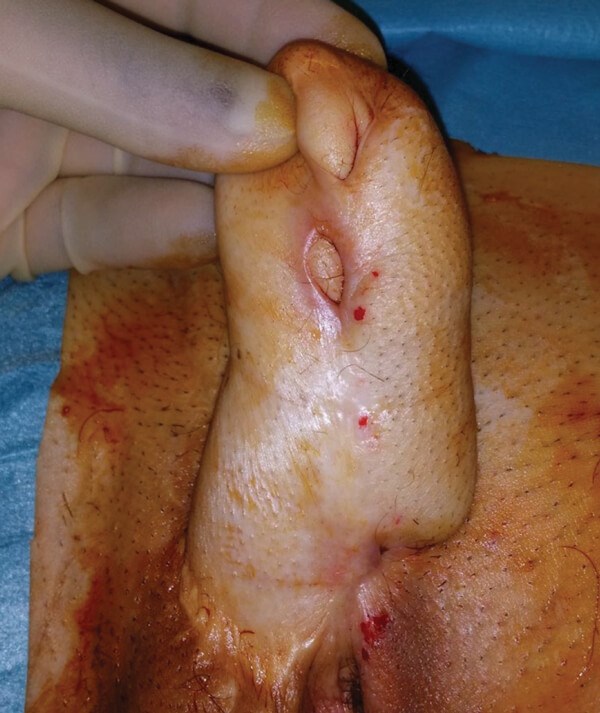
Figure 2: Urethrocutaneous fistula of phalloplasty.
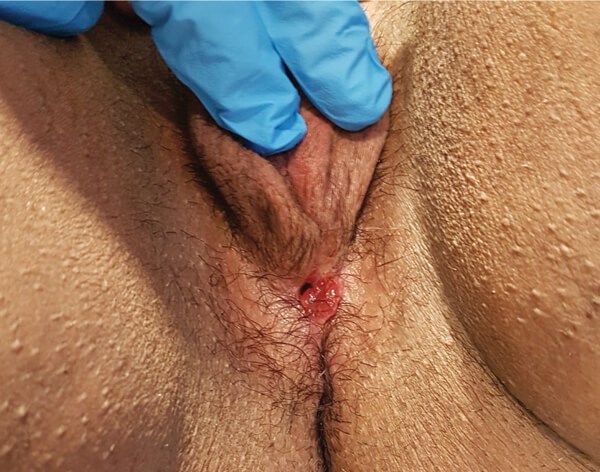
Figure 3: Perineal urethrocutaneous fistula.
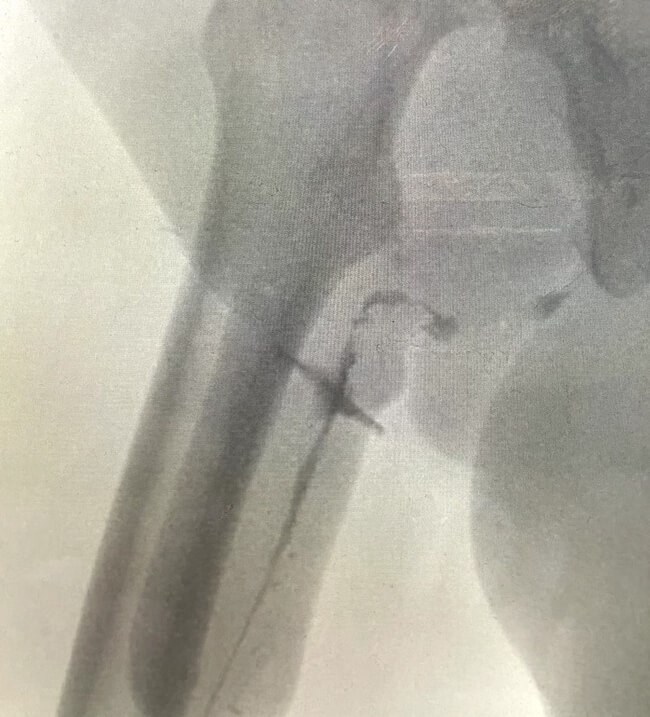
Figure 4: Urethrogram showing a urethrocutaneous fistula.
The most common urethral complication after a phalloplasty is a urethrocutaneous fistula (Figure 2 and 3) with an incidence of 15-70% [6]. Assessment of concomitant urethral strictures is mandatory in these patients (Figure 4). The most common sites for strictures are at the anastomotic sites in phalloplasty or metoidioplasty. The management is more challenging than for individuals assigned male at birth due to the more tenuous blood supply and lack of a corpus spongiosum. Fistulae tend to occur along previous suture lines or at anastomotic sites. A referral to the specialist unit for further investigation and management should be done. If the leaks from the fistulae are bothersome for the patient, then an SPC can be inserted. Other indications for SPC insertion include recurrent infection or excoriation of skin.
Post void dribbling
An element of post void dribbling is common following phalloplasty with urethral join-up. The skin tube urethra slowly dilates over time and urine will pool within the urethra. The scrotal urethra is often tortuous with outpouchings that can collect urine during micturition. A urethral stricture should be excluded by uroflowmetry with a post void residual, followed by retrograde (and antegrade) urethrogram if indicated. Patients should be encouraged to ‘milk’ their urethra each time they void to reduce the dribbling. If it persists and remains bothersome, the patient can be referred for consideration of a reduction urethroplasty.
Post-vaginectomy collection and cysts
After ablation vaginectomy, patients can continue to secrete from retained vaginal mucosa, which may cause pelvic discomfort especially during sexual intercourse. A perineal bulge may also be observed. These collections can fistulate into the urethra or through the skin (or both). Radiological drainage is tempting but should not be done unless there are signs of sepsis (rare). Surgical excision of the remnant mucosa and fistulae is best performed while the cavities are still distended. It will also allow better visualisation by MRI scan.
Urological care following genital gender affirmation surgery
Access to the lower tract (urethra and bladder)
- Retrograde access
It is advisable to carefully use a flexible cystoscope with a guidewire and plenty of lubrication to access the bladder for catheterisation or visualisation. If access is difficult, a perineal urethrostomy can be formed especially if recurrent access with larger sheaths is needed such as for bladder tumour resections and follow-up.- - Anterograde access
Where anterograde access to the bladder is required, care must be taken during bladder puncture and dilation to avoid the vascular pedicle of the phallus. The operating surgeon should discuss with the specialist unit and review the operation records since any pedicle injury will likely result in loss of the phallus.
Access to the upper tracts (kidneys and ureters)
Renal colic or haematuria still need to be investigated as usual noting the advice above concerning retrograde access to the bladder and kidneys for endoscopic procedures. If access to the upper tracts is required, then flexible retrograde ureteroscopy may be possible but rigid retrograde ureteroscopy is rarely possible. However, percutaneous access may be the easier and preferred option.
Lower urinary tract symptoms
De novo overactive bladder syndrome and stress urinary incontinence has been reported following vaginectomy and closure of the vaginal introitus [6,7]. This is necessary for a ‘male-like’ perineum.
Urodynamic assessment should be undertaken at a tertiary centre like University College Hospital in London. The lines can be difficult to insert due to the tortuous urethra and may need to be inserted via a suprapubic approach. Assessment of the fluoroscopic images also require understanding of the anatomy following genital GAS.
Recurrent UTIs
Recurrent UTIs may be a harbinger for the presence of a foreign body in the urethra or bladder. The most common foreign bodies found are hair or stones in the bladder or urethra. These are caused by the use of hair-bearing skin for urethroplasty. The treatment is to remove the stone with an endoscopic stone extraction basket or the use of a laser. Rarely, the stone may require open extraction.
For hairballs located in the penile urethra an artery clip or endoscopic cold cup biopsy forceps can be used for extraction. Depilatory cream can be introduced into the urethra with care and promptly washed out by the patient voiding. To prevent this problem, permanent laser hair removal should be undertaken prior to genital GAS surgery.
Other considerations
Clinicians should be aware that anatomical landmarks may not be as expected following genital GAS. Depending on the surgeries performed the patient’s ovaries, uterus or vagina may still be present. This should be considered if a patient presents with atypical symptoms where the risk of malignancy should not be dismissed despite gender affirmation hormone therapy.
Care should be taken if surgical procedures such as inguinal hernia repair, laparotomy or renal transplantation if required following genital GAS. Referral to the specialist unit will be required if erectile device components are damaged or become infected.
Conclusion
Genital GAS for individuals assigned female at birth is complex and is associated with a significant risk of complications. Most complications are managed by the specialist unit because patients are kept under close follow-up after surgery. However, there will be times when an unwell patient is not able to attend the units in London and will require urgent care at their local hospital. The specialist units can be easily contacted for advice and are happy to support their urological colleagues.
Notwithstanding the complications relating to genital GAS, changes to the anatomical landmarks of individuals can complicate future urological care. A urologist should have a general understanding of the techniques involved in genital GAS. As always, shared care between the local and specialist units will ultimately benefit transgender and non-binary individuals by facilitating care closer to an individual’s home and support network.
TAKE HOME MESSAGE
-
Have a low threshold to contact the specialist unit to discuss the care of individuals assigned female at birth following genital GAS.
-
Urological complications such as urethrocutaneous fistulae and strictures are common after genital GAS.
-
Consider the anatomical changes following genital GAS and the presence of retained structures when planning surgery.
-
Retrograde bladder access should be gentle and performed with the aid of a guidewire and liberal amounts of lubrication.
-
Antegrade access to the bladder must be done carefully, after identifying the location of the vascular pedicle to avoid potential loss of the neophallus.
-
All infected or eroded implants require urgent transfer to the specialist unit for management after starting broad-spectrum intravenous antibiotics.
References
1. Lee WG. The role of penile reconstruction in gender affirmation surgery. Trends in Urology and Men’s Health 2021;12(6):11-14.
2. Van de Grift TC, Elaut E, Cerwenka SC, et al. Surgical satisfaction, quality of life, and their association after gender-affirming surgery: a follow-up study. Journal of Sex and Martial Therapy 2018;44:138-48.
3. Falcone M, Garatta G, Gillo A, et al. Outcomes of inflatable penile prosthesis insertion in 247 patients completing female to male gender reassignment surgery. BJU International 2018;121:139-44.
4. Lee WG, Li V, Ralph DJ, Christopher AN. Novel classification of urethral complications in female to male gender affirmation surgery. Journal of Urology 2021;206(Supplement 3):e542.
5. Lee WG, Christopher AN, Ralph DJ. Gender affirmation surgery, transmasculine. Urological Clinics of North America 2022;49(2):453-65.
6. Fascelli M, Sajadi KP, Digi DD, Geolani WD. Urinary symptoms after genital gender-affirming penile construction, urethral lengthening and vaginectomy. Translational Andrology and Urology 2023;12(5):932-43.
7. Combaz N, Kuhn A. Long-term urogynecological complications after sex reassignment study of 44 patients and diagnostic algorithm proposal. American Journal of Urology Research 2017;2:38‑43.
Declaration of competing interests: None declared.
Aurelie Schirmann and Matthew Megson contributed equally to this article.