Introduction
Disorders of ejaculation are a rare and poorly understood subsection of male sexual dysfunction. A paucity of evidence has hindered advances in definitions, epidemiology, pathophysiology and management. The licensing of a specific medication for premature ejaculation signalled the research interest of pharmaceutical companies in this group of conditions. Furthermore, patients and clinicians are more aware of issues regarding sexual dysfunction. As a consequence a number of new conditions and syndromes have become recognised in the medical literature. Post-orgasmic illness syndrome (POIS) is one such condition.
In 2002 Waldinger and Schweitzer described in a report, two physically healthy men, who developed flu-like symptoms almost immediately after every ejaculation; symptoms would last between two and seven days [1]. Subsequently a limited number of case reports and small series have been published worldwide (approximately 50 in 10 years) [2]. POIS has been recognised as a rare disorder by the National Institute for Health (NIH), Office of Rare Disease Research [3].
Presentation
In Waldinger and Schweitzer’s first report both patients described the rapid onset (30-60 minutes) after ejaculation of feverishness, nasal congestion, itching eyes, fatigue, painful heavy muscles, concentration difficulties and irritation [1]. The symptoms appear to be independent of how the ejaculation is achieved (i.e. masturbation, penetrative intercourse or night-time spontaneous emission) and absent when sexual activity has occurred but ejaculation has not.
Symptoms last between five and seven days and spontaneously resolve until the next ejaculation [4]. The effects are significantly intrusive to lead to abstinence in many men [4]. This is compounded by an association with premature ejaculation (PE). In a cohort of 45 men with POIS, 56% reported having life-long premature ejaculation as defined by the International Society of Sexual Medicine (ISSM) and 15 had an intravaginal ejaculation latency time (IELT) of less than 30 seconds [4]. When compared with the general population the relative risk of males with POIS having PE is 22.4. Whether this is attributable to the avoidance of intercourse, disturbed urogenital function or an unidentified immunogenic / allergic phenomenon is yet to be seen.
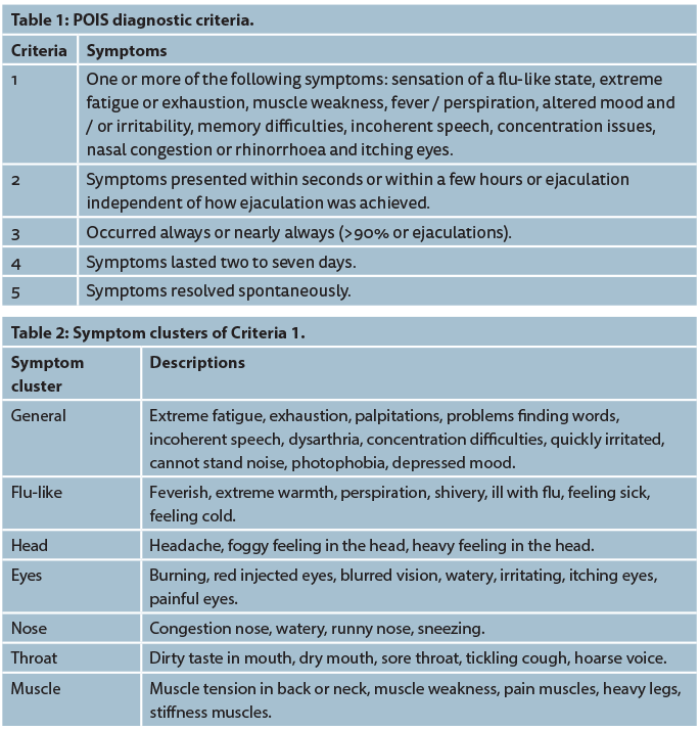
The consequences of POIS on the patient and the partner are significant with many expressing feelings of guilt and concern regarding their sexual relationships [4]. In 2011 Waldinger et al. proposed five preliminary criteria for diagnosing POIS, based on the presentations of 12 patients (Table 1) [4]. A larger cohort of 45 men then validated this.
The symptoms described in the first criteria are quite varied; however, Waldinger et al. went further to cluster them into seven groups as expressed in the patients’ own words (Table 2) [4].
Classification
There appears to be two distinct categories of POIS; primary POIS, in which the symptoms are present from the first ejaculation in puberty (49%); and secondary POIS in which symptoms appear later in life (51%) [4].
Aetiology
A number of theories exist in regard to the underlying pathophysiology of POIS, including aberrant cytokine or neuroendocrine response, and a lack of progesterone [5,6]. The most prevailing of these is that it is due to an allergic / immune mediated response.
Waldinger theorised that the symptom complex of POIS would fit best with a hypersensitivity reaction to seminal fluid. An allergy to human seminal plasma is rare and has been documented in females as a type I hypersensitivity reaction to proteins in the partner’s seminal plasma [7,8].
There are five known types of hypersensitivity reactions; type I is an immediate response to an allergen mediated by IgE; type II is a cytotoxic antibody-dependent reaction mediated by IgM or IgG and complement; type III refers to disorders mediated by IgG and complement; type IV is a delayed hypersensitivity reaction controlled by T-cell-mediated disorders that are antibody independent; and type V autoimmune receptor reactions are disorders that are antibody / complement activation dependent [9].
In the case of POIS the sudden onset of symptoms and local erythema would indicate a type I reaction. The flu-like illness is similar to that seen in type IV hypersensitivity reactions, which manifest in a delayed fashion. Waldinger’s cohort of patients described a peak of severity of symptoms between 24 and 72 hours [4].
Waldinger tested his immune modulated theory by skin-prick testing 33 patients that fulfilled the diagnostic criterion previously set, with samples of autologous semen diluted with 0.9% saline to a concentration of 1:40,000 and compared them with a placebo that was administered to the left arm (0.9% sodium chloride) [4]. An extremely low concentration was used to reduce the potential risk of anaphylaxis. Skin reactions were noted at 15 minutes and graded. A positive skin reaction test was considered when the resultant wheal was >5mm in diameter with accompanying erythema. In the 33 men that underwent testing 29 (88%) had a positive skin prick test compared with 0/33 to 0.9% saline [4]. However, the study acknowledges that it is limited by the absence of a skin prick test of autologous semen in the control group. Waldinger went on to measure the IgE levels in those patients without co-existing allergies and determined that the levels were not raised, thereby calling into question the validity of a type I mediated hypersensitivity reaction being responsible.
More recently Jiang et al. performed a skin-prick test with autologous semen in a patient with POIS and three healthy controls [2]. Positive and negative controls were used (0.1mg/mL histamine and 0.9% sodium chloride) [2]. A skin reaction to autologous semen injection (1:10 and 1:100 dilution) was seen in the POIS patient and the three controls [2]. Interestingly, the responses in the control groups were more pronounced than in the affected individual. There was no detectable increase in serum concentrations of IgE antibodies against seminal fluid when compared with three common allergens in the affected patient [2]. Furthermore, there were no IgE-binding bands on Western blotting and no semen-specific IgE on enzyme-linked immunosorbent assay (ELISA) [2].
The current theory is based on an autoimmune disorder whereby constituents of semen or urethral signalling peptides come into contact with the mucosal lining of the urinary tract and are recognised by dendritic cells that in turn activate naïve T-cells with the seminal fluid antigen, consequently initiating an immune reaction [4,10]. The antigen responsible has yet to be identified, however Waldinger has reported on three men with POIS that persisted after sterilisation suggesting it is likely within the seminal fluid and not bound to the spermatozoa [10].
Treatment
In 1911 Noon and Freedman subjected patients with hay fever to increasing doses of the allergen until resolution of their symptoms [11]. The technique is known as hyposensitisation. The repeated inoculation of the offending allergen leads to the creation of blocking antibodies that combine with the allergen to prevent binding.
Waldinger et al. treated two patients with positive skin prick tests that fulfilled the criteria for POIS with repeated exposure to diluted autologous semen for 31 and 15 months each [12]. Patients received injections 0.05mls of autologous semen at a concentration of 1 in 40,000 diluted with 0.9% sodium chloride [12]. The concentrations were titrated depending on the extent of the skin reaction at 15 minutes. The reaction was graded from 1+ to 5+ [12,13]. The investigators aimed to get a response of 3+. Concentrations were increased when the reaction was ≤2+. If the reaction was >3+ then the concentration were reduced. Where systemic effects were observed the concentration was drastically reduced. Patients received injections fortnightly for one year and then monthly thereafter. The response to treatment was assessed by a non-validated questionnaire giving a score from 0 to 10 with 10 being the worst POIS attack [12].
During the programme, there was a gradual reduction of complaints resulting in 60% and 90% amelioration of POIS complaints at 31 and 15 months, respectively, which coincided in one male with a delay of the IELT from 20 seconds at baseline to 10 minutes after three years of treatment [12].
Alternative treatment options are the use of selective serotonin uptake inhibitors (SSRIs), antihistamines and benzodiazepines [14].
Conclusion
POIS is a rare and debilitating syndrome. Advances have been made in our understanding of the underlying aetiology and an autoimmune response to the patient’s own seminal plasma seems to be the most persuasive argument. The specific mechanism of the observed IELT with POIS remains unknown and needs to be elucidated. A number of strategies have been trialled to treat this disorder, however success cannot be guaranteed and patients should be counselled appropriately to be made aware of the low-level of evidence available.
References
1. Waldinger MD, Schweitzer DH. Postorgasmic illness syndrome: two cases. J Sex Marital Ther 2002;28(3):251-5.
2. Jiang N, Xi G, Li H, Yin J. Postorgasmic illness syndrome (POIS) in a Chinese man: no proof for IgE-mediated allergy to semen. J Sex Med 2015;12(3):840-5.
3. National Institute for Health (NIH). Postorgasmic illness syndrome. 2016.
https://rarediseases.info.nih.gov/
diseases/10809/postorgasmic-illness-syndrome
Accessed February 2017.
4. Waldinger MD, Meinardi MM, Zwinderman AH, Schweitzer DH. Postorgasmic Illness Syndrome (POIS) in 45 Dutch caucasian males: clinical characteristics and evidence for an immunogenic pathogenesis (Part 1). J Sex Med 2011;8(4):1164-70.
5. Dexter S. Benign coital headache relieved by partner’s pregnancies with implications for future treatment. BMJ case reports 2010;2010.
6. Ashby J, Goldmeier D. Postorgasm illness syndrome - a spectrum of illnesses. J Sex Med 2010;7(5):1976-81.
7. Ohman JL Jr, Malkiel S, Lewis S, Lorusso JR. Allergy to human seminal fluid: characterization of the allergen and experience with immunotherapy. J Allergy Clin Immunol 1990;85(1 Pt 1):103-7.
8. Lee J, Kim S, Kim M, et al. Anaphylaxis to husband’s seminal plasma and treatment by local desensitization. Clin Mol Allergy 2008;6:13.
9. Adkins NF, Friedman PS, Pongracic JA. Drug allergy. In: Holgate ST, Church MK, Lichtenstein LM. Allergy. Philadelphia, USA; Mosby Elsevier; 2006: 157-66.
10. Waldinger MD. Post orgasmic illness syndrome (POIS). Transl Androl Urol 2016;5(4):602-6.
11. Noon L. Prophylactic inoculation against hay fever. The Lancet 1911;177:1572-3.
12. Waldinger MD, Meinardi MM, Schweitzer DH. Hyposensitization therapy with autologous semen in two Dutch caucasian males: beneficial effects in Postorgasmic Illness Syndrome (POIS; Part 2). J Sex Med 2011;8(4):1171-6.
13. Koller DY, Pirker C, Jarisch R, Gotz M. Influence of the histamine control on skin reactivity in skin testing. Allergy 1992;47(1):58-9.
14. Althof SE, McMahon CG. Contemporary Management of Disorders of Male Orgasm and Ejaculation. Urology 2016;93:9-21.




