Urinary incontinence is a common condition faced by up to 20-50% of women, which impairs quality of life and poses a significant socioeconomic burden to both the individual women involved and more widely to the NHS [1]. The overall cost of stress urinary incontinence (SUI) alone to the NHS has been reported at over £117 million pounds per year [2]. Ultimately women have a 10% lifetime risk of requiring continence surgery.
However, the management of SUI has changed dramatically over the past few years [3]. The current UK wide mesh suspension, along with the Supreme Court ruling on the Montgomery vs. Lanarkshire legal case have heavily influenced the way in which we counsel and consent patients in all aspects of their care, not just for surgical intervention [4].
Many patients have been involved in the litigation process against the use of vaginal mesh on the basis of what was felt to be a lack of informed consent at the time of surgery about the potential future complications. Consequent to a paternalistic healthcare system, women specifically feel they have been failed in the pre-operative clinic setting when they have been counseled on the surgical options available.
“it is essential that these women embrace an active role in their healthcare and understand the implications any surgery may have on their existing and future lifestyle, fitness and social life”
Procedural data retrospectively collected from within the NHS has illustrated that between 2008 and 2017 over 100,516 vaginal meshes were inserted whilst only 1195 non-tape SUI procedures were performed [1].
The move towards shared decision-making
Given its extensive involvement in the current mesh surgery pause now in place throughout the UK, NHS Scotland has increasingly become the driving force in the changing process of consent and decision- making for women undergoing SUI surgery. ‘Shared decision-making’ is one of the six key pillars in the concept of ‘Realistic Medicine’ which was introduced by the Scottish Government in 2016 by the Chief Medical Officer [5]. The concept of shared decision-making is one that combines the medical knowledge of the clinician and the personal preferences of the patient to derive an appropriate plan for treatment. It moves away from a unilateral delivery of information to patients towards a more active participation from patients with support from healthcare professionals in the decision-making process [6].
The reasons behind a continence PDA
In the light of the current mesh controversies and along with the introduction of shared decision-making, whilst striving to deliver ‘patient-centred care’, the idea of a continence patient decision aid (PDA) was formulated within NHS Ayrshire & Arran (NHS A&A). The intention was to help women work through and document their decision-making processes when contemplating surgery.
Women were encouraged to discuss and record their thoughts for both choosing and rejecting a specific treatment option after the provision of verbal and written information on each type of treatment. The aim was to make the women feel they are a key part of the decision-making process rather than being a passive recipient of information and advice from clinicians about which surgery they should have, as has traditionally been the situation. By engaging with women to take part in the process, we strongly believe it empowers them and promotes ownership of personal healthcare decisions.
The PDA consultant process
Within NHS Ayrshire & Arran, there is a well-defined pathway for the treatment of SUI. Once patients have completed conservative measures under supervision from our specialist nurse-led continence service and are at the point that they wish to consider some form of operative intervention, they are introduced to the potential treatment options by our specialist nurses. As part of the discussion they are provided with patient information leaflets on intra-urethral bulking injections, colposuspension (open and laparoscopic) and natural tissue sling – either from the British Association of Urological Surgeons (BAUS) or British Society of Urogynaecology (BSUG) and a copy of the NHS A&A PDA paperwork.
Women are initially introduced to and taken through the procedures available (excluding mesh at present) by one of our nurse specialists and the PDA form is explained to them with advice on how to complete it. They then have an outpatient appointment made with either a consultant urologist or uro-gynaecologist.
At this consultation women are further counselled about the technical aspects of the individual operations, success rates, risks, benefits and alternatives, typical recovery and length of stay. They are encouraged to ask questions and discuss any concerns they may have based on their initial reading and conversations with the nurse specialists. They are specifically encouraged to highlight worries that they themselves find personally troubling or potentially concerning, such as restriction of activities postoperatively, time off work or the need to self-catheterise. The women are then required to complete the PDA in their own time, once they have absorbed and assimilated the information provided from all sources and return their paperwork to the department for discussion via the incontinence multidisciplinary meeting (MDT). If women are still struggling with their choice, a further outpatient appointment is provided.
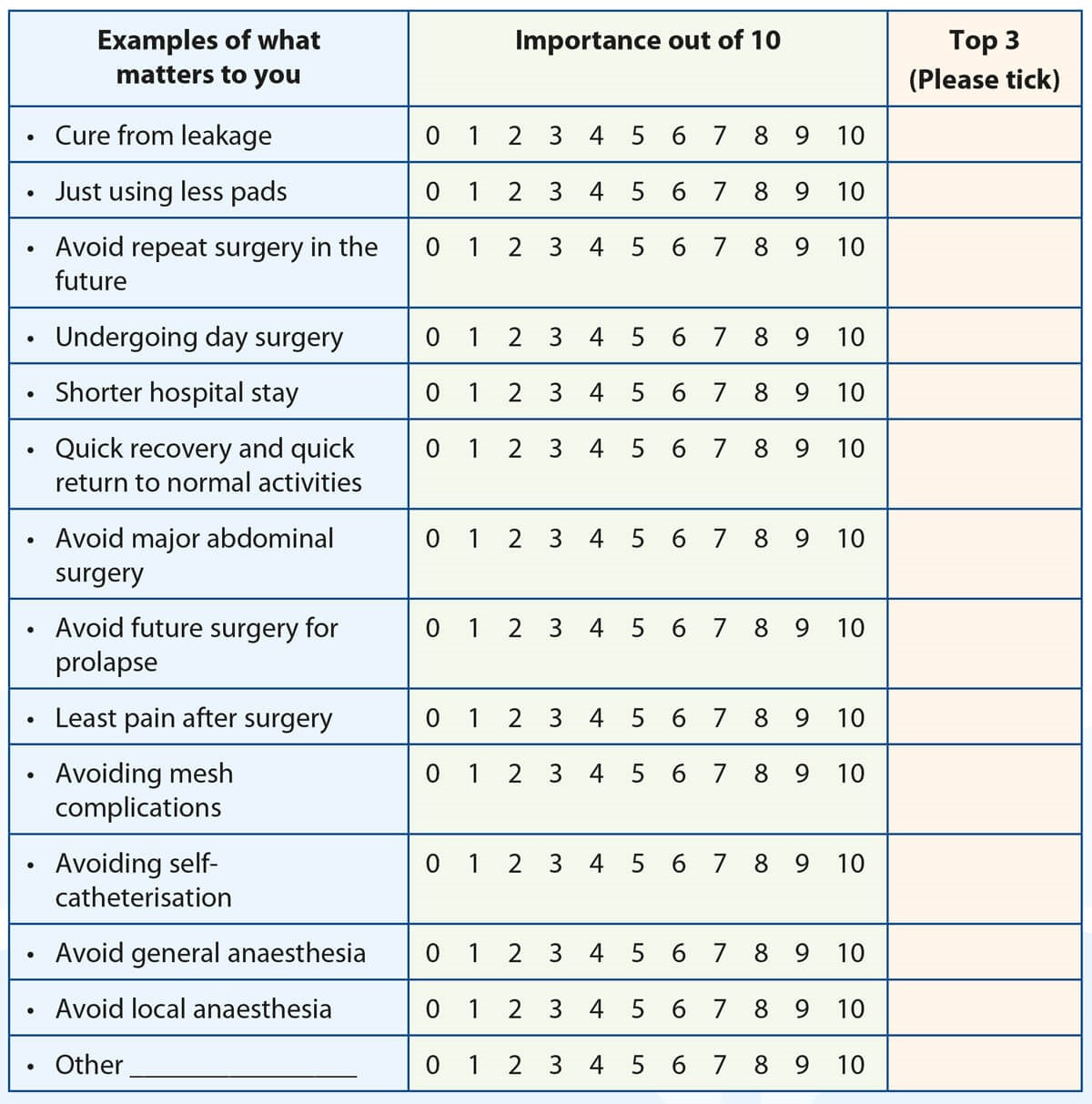
Figure 1.
The PDA document explained
The PDA developed by NHS A&A contains several different components that aid women in documenting their thought process for undergoing SUI surgery. It is a 12-page document issued to patients contemplating intervention for their incontinence. The fundamental idea behind the PDA is to help women identify ‘What Matters To Them’ and supports them to make a decision that is best for them. The first section (Figure 1) asks women to identify their values when dealing with urinary incontinence. Thirteen statements such as: ‘cure from leakage’, ‘using less pads’, ‘avoiding repeat surgery’, ’avoiding major surgery’, ‘avoid mesh’ or ‘quick recovery’ are rated in importance from 0-10.
Women are then asked to highlight the top 3 that matter the most to them. The reason for asking patients to document their thought process is that it allows the MDT to identify what is important to women in their motivation for choosing (or discounting) particular surgical options. Women are then encouraged to think about the physical, psychological and social implications of their decision. A flow chart is provided to re-cap non-surgical options for patients in order for them to feel confident that they are ready to consider surgical intervention as the next step in the management of their incontinence.
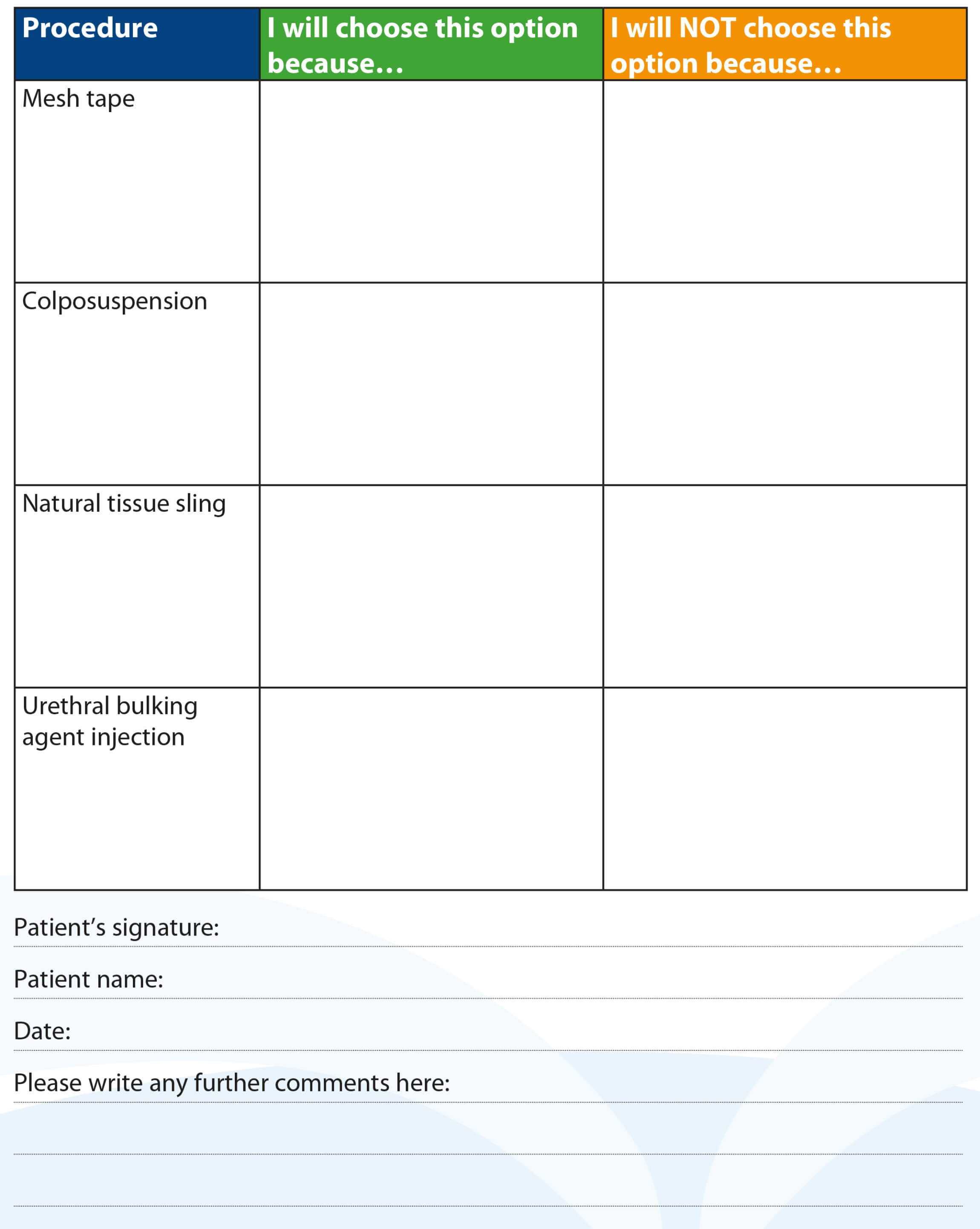
Figure 2.
The next step within the PDA is for patients to review a table outlining the main advantages, disadvantages and implications of vaginal tape surgery, colposuspension, natural tissue sling and urethral bulking agents. This summarises the information that has been provided throughout the process about these different types of surgeries, although all women are informed that mesh is currently unavailable within the UK. Once the women have processed all the information given to them, they are then asked to fill in the table indicating their choice (Figure 2). For each procedure they are requested to document why they would or would not choose each of the four and then sign the form to confirm their choice.
The final component of the PDA is the decisional conflict scale, which was not part of the original concept, but was an addition in an updated version to allow patients to document the ease with which they made their decision. This scale allows the MDT to gauge whether patients understand their choice or in fact need more support in their decision with further consultation and discussion about their options.
Benefits and potential problems
As expected with all tools for aiding the delivery of patient care and engagement in treatment decision, there are benefits and limitations that need to be acknowledged. Happily, many women have reported that they find the PDA particularly helpful because “all risks, not just what medics feel are important” are discussed and furthermore women “appreciate open conversations”. The PDA has been described as “easy to use and made me think about what is really important to me” and the entire process has been praised by women who state “this is great, makes me feel that you want to take the time to know me”.
However, the use of the PDA undoubtedly raises issues – especially regarding patient literacy, understanding or managing those women who do not want to make decisions themselves. Some women simply wish to be told what operation they should have and do not want to be informed of risks or complications that may arise.
Our concern has been that, as demonstrated by the Montgomery vs. Lanarkshire case, the importance and value of risk varies between patients. What is acceptable to one person may be entirely unacceptable to others and therefore by having open, frank and extensive consultations with women to be able to understand what matters significantly for them is a key part of the process. By documenting the reasons for their particular surgical choice, thereby ensuring the women are as informed and educated as possible, future dissatisfaction is potentially minimised.
Another concern raised has been that some women will simply complete the PDA using phrases that will get them the operation that they want, based on opinions of others. Whilst this is always a potential risk, it is essential that these women embrace an active role in their healthcare and understand the implications any surgery may have on their existing and future lifestyle, fitness and social life.
Use of our PDA has raised a few instances where patient choice has significantly differed from the decision of the treating clinician and the MDT process after formal assessment of the SUI. These women are subsequently reviewed back in the outpatient clinic to facilitate further discussion about surgery. Detailed explanations are given as to why a particular intervention may not be suitable or less successful and alternative treatment suggestions explored.
The most common reason for discordance is patient fitness and often requires input from our anaesthetic colleagues to highlight unacceptable operative risks that may occur from surgery. One of the major findings we have established from analysing responses on the PDA is that women are actively choosing procedures that are less invasive. Many women are not ready to commit to major surgery and so are willing to accept a reduced efficacy from a more minimal intervention, over more effective major surgery with a longer recovery and increased risks.
Audit
The MDT audited patients who had completed the PDA to see whether it impacted upon their decision when choosing SUI surgery [7]. The handwritten free text of the decision table underwent content theme analysis and identified ‘efficacy’, ‘safety’, ‘invasiveness’, ‘recovery’ and ‘blank’ as the most common themes documented on the PDA by women in their decision process.
In the 18 months prior to the introduction of the PDA: 19 women underwent colposuspension; 44 had intra-urethral injections in the form of Bulkamid; and 0 patients had a natural tissue sling in NHS A&A. No patients were offered a vaginal mesh procedure at that time, as the mesh suspension was in effect within Scotland.
The PDA was then introduced in September 2016 and the responses of 30 women were studied over an 18-month period. Twenty-four women selected a colposuspension and three a fascial sling for ‘efficacy’ and five requested bulking injections because they were the ‘least invasive’. Despite not being available, one patient wished a vaginal mesh for the specific reason of ‘recovery’. ‘Cure from SUI’ was identified as one of the top three values in 60% of women requesting bulking injections, 63.6% of colposuspension and 66.6% of sling requests. Vaginal mesh was rejected for ‘safety concerns’ by 24/30 women. Colposuspension was rejected by four women for concerns about prolapse, by two for level of ‘invasiveness’, by one due to ‘efficacy’ and by one for ‘recovery’ reasons. Thirteen women rejected slings due to ‘safety concerns’ or the need to catheterise and 17 patients excluded bulking for ‘efficacy’.
Conclusion
Ultimately, by introducing the PDA we have been able to increase our understanding of the patient values behind their thought process for choosing a particular surgery. It is helpful when counselling women to understand what worries them and allows clinicians to focus consultations on ensuring these are addressed clearly. By having the addition of a decision conflict scale, this helps highlights women who may have struggled to come to their decision. Clinicians can then provide further opportunities to make sure women are entirely happy and understanding of their choice of continence surgery.
We believe the PDA is an easily adaptable tool for other units to implement to help women navigate through the difficult field of continence surgery and we welcome the NICE produced tool. The NHS A&A PDA has recently been validated in our population within the West of Scotland and would be of value for other units undertaking such procedures to assist both women and clinicians make the best choice for their healthcare needs [8].
References
1. Abrams P, Cardozo L, Khoury S, Wein A (Eds.). Incontinence, 5th Edition: International Conference on Incontinence, Paris, February 2012. ICUD-EAU; 2013.
2. Papanicolaou S, Pons ME, Hampel C, et al. Medical resource utilisation and cost of care for women seeking treatment for urinary incontinence in an outpatient setting. Maturitas 2005;52(Suppl. 2):S35-47.
3. Abrams P, Cardozo L, Wagg A, Wein A (Eds.). Incontinence 5th Edition (2013). ICI-ICS. International Continence Society; 2013.
4. https://www.supremecourt.uk/
cases/docs/uksc-2013-0136-judgment.pdf
Accessed 22 January 2020.
5. https://www2.gov.scot/
resource/0049/00492520.pdf
Accessed 22 January 2020.
6. https://www.gov.scot/publications/
works-support-promote-shared
-decision-making-synthesis
-recent-evidence/
Accessed 22 January 2020
7. Spyroulis C, Sokolova I, Bekarma H, et al. Does the use of patient decision aid (PDA) influence the patterns of surgery for stress urinary incontinence? Abstract EUGA; 2018.
8. Ong HL, Sokolova I, Bekarma H, et al. Development, validation and initial evaluation of patient-decision aid (SUI-PDA©) for women considering stress urinary incontinence surgery. Int Urogynecol J 2019;30:2013-22.
Declaration of competing interests: None declared.