Testicular cancer (TC) is the most successfully treated solid tumour, achieving a cure rate of 90-95% [1-3]. Testicular cancer is relatively rare with an incidence of 2207 cases in the UK in 2014 [4] and yet is the most common cancer in men aged 15-49. The incidence of testicular cancer is increasing, with a 6% increase in incidence in 2009-2011 compared to 2000-2002 [4]. Occupational, lifestyle and socioeconomic factors have been suggested as putative causes, however no definitive cause for the increased incidence has been identified.
Aetiology
Although a definitive cause for testicular cancer has never been identified, several risk factors are known. These include cryptorchidism, carcinoma in situ (intratubular germ cell neoplasia), prior history of testis cancer or extragonadal germ cell tumour and positive family history (first degree relatives) [5].
Classification
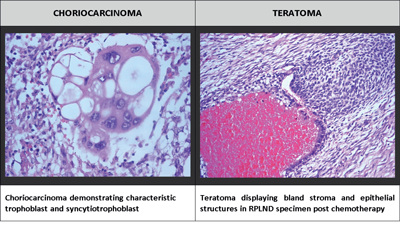
Germ cell tumours (GCT) are divided into seminomas and non-seminomatous GCTs. Non-seminomas contain several histological subtypes (including choriocarcinoma, embryonal carcinoma, yolk-sac tumour and teratoma), which are grouped together. Non-seminomas may contain one histological subtype or a mixture of two or more subtypes. Among GCTs approximately 44% are non-seminomas [6].
Presentation and diagnosis
Diagnosis is based on history, examination, tumour markers, testicular ultrasound and staging CT scan. Definitive diagnosis is established by histopathological analysis of tissue from the orchidectomy specimen. Men typically present with a unilateral testicular mass that is usually painless. However, up to 20% of men complain of pain, testicular discomfort or heaviness.
Tumour markers for GCT include alpha-fetoprotein (AFP), beta human chorionic gonadotropin (ß-hCG) and lactate dehydrogenase (LDH). AFP and ß-hCG are specific and sensitive markers and play a role in both the diagnosis and assessing prognosis. It is essential to check tumour markers both pre and post-surgery. Persistently raised tumour markers post-orchidectomy suggests the presence of metastatic disease and should be treated as such [2].
Testicular ultrasound is the imaging modality of choice to assess a testicular mass or swelling as it is sensitive, inexpensive and non-invasive. A CT scan of the chest, abdomen and pelvis with intravenous contrast to assess the retroperitoneum and mediastinum for pathological lymphadenopathy is standard. Fluorodeoxyglucose-positron emission tomography (FDG-PET) has failed to show any benefit over CT scan for initial staging of TC [3].
Figure 1: Histological subtypes of non-seminomatous cell tumour.
Treatment of primary tumour
Radical inguinal orchidectomy is the standard surgical treatment. Testicle-preserving surgery is a consideration in specialised centres in cases of synchronous bilateral testicular tumours, metachronous contralateral (second) testicular tumour, tumour in a solitary testis, and where there is a contralateral atrophic testis [2].
Adjuvant treatment
Treatment options for stage 1 non-seminoma, post-orchidectomy include surveillance, adjuvant chemotherapy and nerve sparing retroperitoneal lymph node dissection (RPLND). The presence of vascular invasion (VI) classifies patients into low-risk (VI negative) or high-risk (VI positive) groups with five-year relapse rates of 20% and 40-50%, respectively. Both groups are associated with excellent survival rates of greater than 95%.
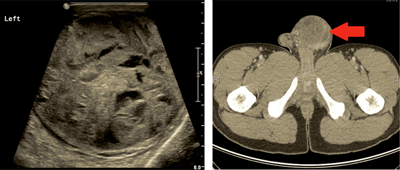
Figure 2: Ultrasound images demonstrating an enlarged left testicle with presence of heterogeneous mass which is re-demonstrated on axial cross-sections of an abdomen-pelvic CT scan in a patient with non-seminoma GCT.
Treatment of low-risk stage 1 non-seminoma
Low-risk patients are offered surveillance as a standard option unless follow-up is not possible due to lack of surveillance facilities or patient choice, in which case treatment with one or two cycles of Bleomycin, Etoposide, Cisplatin (BEP) is recommended. Overall 20% of patients will relapse, with the majority (95%) of relapses occurring over the first two years. The European Society of Medical Oncology (ESMO) surveillance schedule recommends frequent monitoring during the initial two years (Table 1).

Surveillance provides a means to avoid over-treatment with chemotherapy – however, it requires an experienced clinical team and a compliant patient group to be effective. The surveillance process is often accompanied by anxiety and psychological stress. It is important for the medical team to consider social and emotional aspects of a patient’s care whilst on surveillance.
Treatment of high-risk stage 1 non-seminoma
The standard treatment for high-risk stage 1 non-seminoma is either surveillance or one or two cycles of adjuvant chemotherapy with BEP. The decision is based on discussion with patients while providing them with detailed information about risks and benefits of each approach.
The relapse rate on surveillance is approximately 40-50%, which indicates that 50% of patients will eventually receive chemotherapy. Treatment with two cycles of BEP reduces the relapse rate to 3-4%. It is important to note that the survival remains approximately 95% in both treatment groups. A concern with offering surveillance to patients in the high-risk group, is that in the event of disease relapse, an RPLND on completion of chemotherapy may be required. In the event of relapse, treatment is chemotherapy with three to four cycles of BEP.
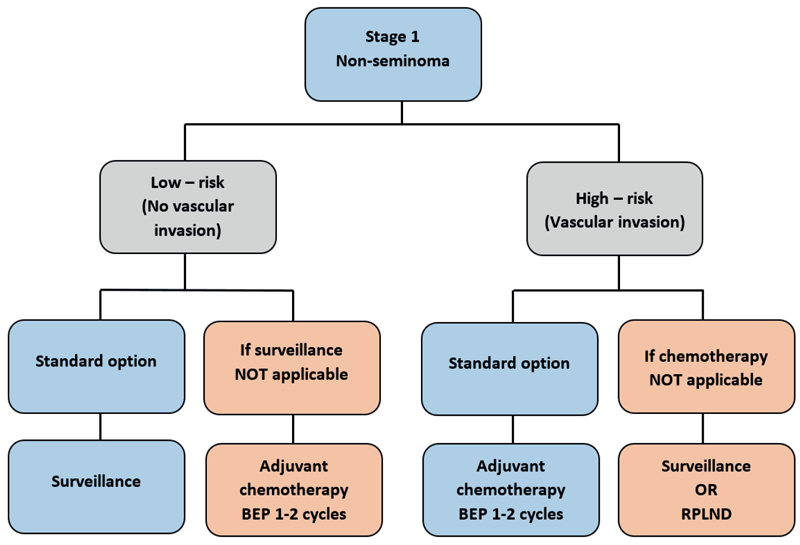
Figure 3: Treatment algorithm for stage 1 non-seminoma.
Considering the long-term toxicities associated with BEP chemotherapy, efforts are being directed towards reducing exposure to chemotherapy. Patients treated with BEP have twice the risk of developing a secondary malignancy, most commonly leukaemia, gastrointestinal cancers, genitourinary cancers, lung cancer and sarcoma. Receiving BEP chemotherapy doubles the risk of developing metabolic syndrome and cardiovascular disease (i.e. congestive heart failure, myocardial infarction, cerebrovascular accidents) [7,8].
In an effort to reduce toxicity different groups have looked at using one cycle of BEP. The Swedish-Norwegian Testicular Cancer Project (SWENOTECA), a large population-based study, which included 745 patients with stage 1 non-seminomatous disease (VI positive) with a median follow-up of 4.7 years, demonstrated a relapse rate of 3.2% in men with stage 1 NSGCT with vascular invasion treated with one cycle of BEP [9]. At present the 111 trial, a single arm, multicentre, UK study is evaluating one cycle of BEP as adjuvant chemotherapy in high-risk, stage 1 NSGCT. The study has recently completed accrual and will be positive if it shows a two-year recurrence rate of less than 5%.
Open, laparoscopic or robot-assisted RPLND in stage 1 non-seminoma is restricted to cases where surveillance or adjuvant chemotherapy are not applicable or there is diagnostic uncertainty. In view of the complex nature of the surgery, RPLND should be performed by specialist surgeons only. The rare absolute indication for RPLND is a >2cm residual lymph node mass on completion of treatment with chemotherapy for advanced (stage II, III) NSGCT and should be considered if complete resection is feasible [2].
Follow-up
Early detection and treatment of relapse represents the primary objective of follow-up visits. Follow-up schedules include physical examination, tumour markers and radiological imaging. Relapses are most commonly detected by marker elevation [1]. As exposure to excessive radiation could cause secondary malignancies, future guidelines are likely to reduce the frequency of CT scans, as the patient population has a long life-expectancy after successful treatment [10].
TAKE HOME MESSAGE
-
Testicular ultrasound is an inexpensive, non-invasive and a sensitive imaging modality in the diagnosis of testicular cancer.
-
The main predictor of relapse in Stage 1 NSGCT is histopathological evidence of vascular invasion.
-
Low-risk stage 1 non-seminoma should be offered surveillance within an experienced surveillance programme.
-
If surveillance is not feasible in low-risk patients, adjuvant chemotherapy or nerve-sparing RPLND are treatment options.
-
Primary chemotherapy with one or two cycles of BEP is recommended for high-risk stage 1 non seminoma.
-
Surveillance or nerve-sparing RPLND in high-risk patients remains an option for those not willing to undergo adjuvant chemotherapy.
Acknowledgement
The images featured in this article were provided by Dr Ashish Chandra Consultant Pathologist, Guy’s and St Thomas’ NHS Foundation Trust.
References
1. de Wit R, Fizazi K. Controversies in the Management of Clinical Stage I Testis Cancer. Journal of Clinical Onology 2006;24(35):5482-92.*
2. Albers P, Albrecht W, Algaba F, et al. European Association of Urology Guidelines on Testicular Cancer. 2014.*
3. Schmoll H-J, Jordan K, Huddart R, et al.; on behalf of the ESMO Guidelines Working Group. Testicular non-seminoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2010;21(5):147-54.*
4. Cancer Research UK. UK Cancer Incidence (2011) by Country Summary. 2014.
5. Garner MJ, Turner MC, Ghadirian P, Krewski D. Epidemiology of testicular cancer: an overview. Int J Cancer 2005;116(3):331-9.
6. McGlynn KA, Cook MB. Etiologic factors in testicular germ-cell tumors. Future Oncol 2009;5(9):1389-402.
7. Willemse PM, Burggraaf J, Hamdy NAT, et al. Prevalence of the metabolic syndrome and cardiovascular disease risk in chemotherapy-treated testicular germ cell tumour survivors. Br J Cancer 2013;109(1):60-7.
8. Beyer J, Albers P, Altena R, et al. Maintaining success, reducing treatment burden, focusing on survivorship: highlights from the third European consensus conference on diagnosis and treatment of germ-cell cancer. Ann Oncol 2013;24:878-88.
9. Tandstad T, Cohn-Cedermark G, Dahl O, et al. Long-term follow-up after risk-adapted treatment in clinical stage 1 (CS1) nonseminomatous germ-cell testicular cancer (NSGCT) implementing adjuvant CVB chemotherapy. A SWENOTECA study. Ann Oncol 2010;21(9):1858-63.
10. Brenner DJ, Hall EJ. Computed tomography: an increasing source of radiation exposure. N Engl J Med 2007;357(22):2277-84.
* Key references.