Case 1
A 78-year-old female presents with a week history of painless haematuria. She’s a smoker and used to work in the textiles industry. She underwent a flexible cystoscopy.
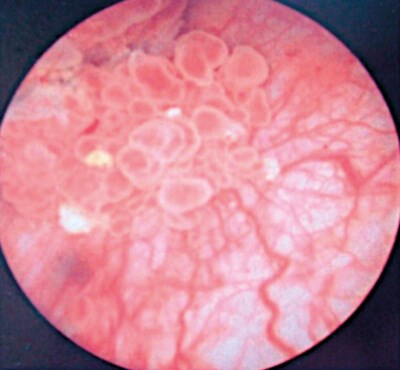
- What does this image show?
- What are the two-week wait National Institute for Health & Care Excellence (NICE) referral criteria for bladder cancer?
- How would you manage this patient?
- The histology showed a G1pTa lesion under 3cm in size. What follow-up would you arrange for this patient?
- What would be your next steps if the histology had shown G3pT1 disease?
Case 2
A 67-year-old male underwent imaging as part of a haematuria work-up. He is otherwise fit and well and is an ex-smoker.
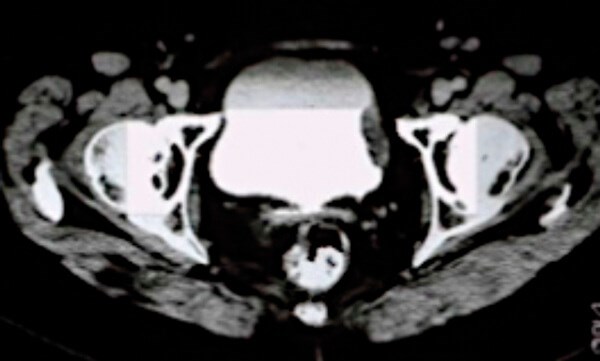
Figure 1.
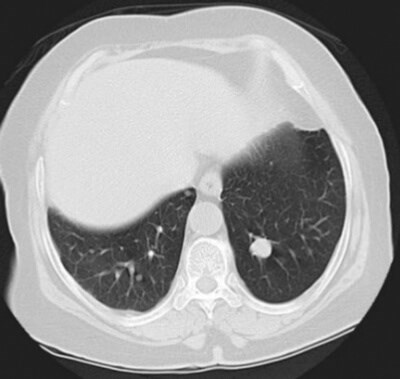
Figure 2.
- What does the scan in Figure 1 show?
- What are the surgical and non-surgical treatment options available to this patient if the histology was in keeping with muscle invasive disease, providing there are no distant metastases?
- What surgical risks would you consent the patient for if he decided to undergo surgery?
- How should this patient be followed up following surgery?
- The patient had the test shown in Figure 2 in clinic as part of their postoperative follow-up. What does it show?
- Considering the image above, what are the management options available?
Bladder cancer: answers
Case 1
1. A <3cm bladder tumour with a papillary appearance.
2. The NICE guidelines recommend referral to urology for individuals aged 45 and over and have:
-
Unexplained visible haematuria without urinary tract infection (UTI), or
-
Visible haematuria that persists or recurs after successful treatment of UTI, or
-
Aged 60 and over and have unexplained non visible haematuria and either dysuria or a raised white cell count on a blood test,
-
Non-urgent referral in subjects aged 60 and over with recurrent or persistent unexplained UTI.
3. This patient should have a transurethral resection of bladder tumour (TURBT) with a single dose of intravesical Mitomycin C immediately after resection. If muscle invasive disease is suspected then CT or MRI staging should be undertaken.
4. This patient has low-risk non-muscle invasive bladder cancer (NMIBC) and should be offered cystoscopic follow-up at 3 and 12 months following diagnosis. If the patient has not developed a recurrence within 12 months then they can be discharged back to primary care.
5. The management here differs as the patient has high risk NMIBC. This patient should undergo a re-resection TURBT. The indications for re-resection are the following:
-
Incomplete initial TURBT or concerns about completeness.
-
No muscle in the specimen after initial resection; the exception is primary carcinoma in situ (CIS) tumours
-
All pT1 tumours.
Case 2
1. This is a CT urogram of the pelvis demonstrating a filling defect in the bladder suspicious for muscle invasive bladder cancer. A CT chest is needed to complete the staging.
2. Surgical option: radical cystectomy with pelvic lymph node dissection and neoadjuvant chemotherapy. Non-surgical option: external beam radiotherapy. 3. Bleeding possibly requiring blood transfusion; Infection (wound, chest, urine or intra-abdominal collections requiring drainage); Visceral injury (including rectal injury possibly necessitating a colostomy); Anastomotic leak (bowel or uretero-ileal); Ileus or bowel obstruction; Anaesthetic or cardiovascular complications (deep vein thrombosis, pulmonary embolism, myocardial infarction, stroke and death); Renal failure; Stoma complications (stenosis, prolapse or hernia requiring revision); Stricturing at bowel or uretero-ileal anastomosis; Incisional hernia; Vitamin and electrolyte abnormalities requiring supplementation (e.g. sodium bicarbonate).
4. CT chest, abdomen and pelvis (CAP) at 6, 12 and 24 months to monitor for local and distant recurrences. Urethral washing for cytology and / or urethroscopy annually for five years to detect urethral recurrences. Monitoring for metabolic acidosis and B12 deficiency and folate deficiency annually. Monitoring upper tracts for hydronephrosis, stones and cancer using imaging and glomerular filtration rate (GFR) estimation at least annually.
5. This is a CT scan image of the chest demonstrating a lesion in the left upper lobe. This lesion would be highly suspicious of tumour recurrence given this patient’s history.
6. Assuming this lesion represents metastatic disease, the management options are dependent on a number of factors: patients’ comorbidities, renal function and performance status. First-line treatment options include platinum-based chemotherapy in patients who are fit. Programmed cell death protein 1 (PD-1) and programmed death ligand 1 (PD-L1) inhibitor immunotherapy treatment can be used in patients who progress on platinum-based treatments or in those who are ineligible for platinum-based first line treatments.